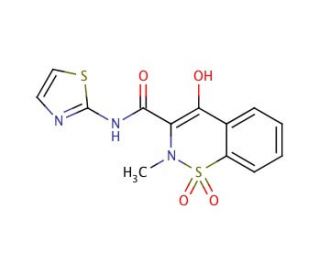
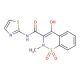
Molecular structure of Sudoxicam, CAS Number: 34042-85-8
Sudoxicam (CAS 34042-85-8)
See product citations (1)
Alternate Names:
4-Hydroxy-2-methyl-N-2-thiazolyl-2H-1,2-benzothiazine-3-carboxamide 1,1-Dioxide; N-(2-Thiazolyl)-4-hydroxy-2-methyl-2H-1,2-benzothiazine-3-carboxamide 1,1-Dioxide
Application:
Sudoxicam is a nonsteroidal, anti-inflammatory drug
CAS Number:
34042-85-8
Molecular Weight:
337.37
Molecular Formula:
C13H11N3O4S2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Sudoxicam that belongs to the oxicam class of compounds. It inhibits the cyclooxygenase (COX) enzymes, which are key in the biosynthesis of prostaglandins The oxicam class is characterized by a specific chemical structure that confers its ability to inhibit COX enzymes, thereby reducing the production of prostaglandins. Sudoxicam is not naturally produced within the body but is introduced externally, being detected in individuals who have been exposed to the compound or its related substances. As such, Sudoxicam is considered a component of the human exposome, which encompasses all exposures an individual encounters throughout their life.
Sudoxicam (CAS 34042-85-8) References
- Metabolism of sudoxicam by the rat, dog, and monkey. | Hobbs, DC. and Twomey, TM. 1977. Drug Metab Dispos. 5: 75-81. PMID: 13979
- In vitro metabolism and covalent binding of enol-carboxamide derivatives and anti-inflammatory agents sudoxicam and meloxicam: insights into the hepatotoxicity of sudoxicam. | Obach, RS., et al. 2008. Chem Res Toxicol. 21: 1890-9. PMID: 18707140
- Idiosyncratic reactions and metabolism of sulfur-containing drugs. | Zuniga, FI., et al. 2012. Expert Opin Drug Metab Toxicol. 8: 467-85. PMID: 22394356
- Dual mechanisms suppress meloxicam bioactivation relative to sudoxicam. | Barnette, DA., et al. 2020. Toxicology. 440: 152478. PMID: 32437779
- Meloxicam methyl group determines enzyme specificity for thiazole bioactivation compared to sudoxicam. | Barnette, DA., et al. 2021. Toxicol Lett. 338: 10-20. PMID: 33253783
- Ultrasensitive and simultaneous detection of 6 nonsteroidal anti-inflammatory drugs by colloidal gold strip sensor. | Lin, L., et al. 2021. J Dairy Sci. 104: 2529-2538. PMID: 33455779
- Improvement of the gastric tolerance of non-steroidal anti-inflammatory drugs by polyene phosphatidylcholine (Phospholipon 100). | Leyck, S., et al. 1985. Eur J Pharmacol. 117: 35-42. PMID: 4085544
- Anti-inflammatory and pharmacokinetic properties of sudoxicam N-(2-thiazolyl)-4-hydroxy-2-methyl-2H-1,2-benzothiazine-3-carboxamide 1,1-dioxide. | Wiseman, EH. and Chiaini, J. 1972. Biochem Pharmacol. 21: 2323-34. PMID: 4630489
- Inhibition of platelet aggregation and of experimental thrombosis by sudoxicam. | Constantine, JW. and Purcell, IM. 1973. J Pharmacol Exp Ther. 187: 653-65. PMID: 4770405
- Interaction of sudoxicam and aspirin in animals and man. | Wiseman, EH., et al. 1975. Clin Pharmacol Ther. 18: 441-8. PMID: 809225
- Anti-inflammatory drugs and renal papillary necrosis. | Wiseman, EH. and Reinert, H. 1975. Agents Actions. 5: 322-5. PMID: 813505
- Pharmacokinetics of oxicam nonsteroidal anti-inflammatory agents. | Olkkola, KT., et al. 1994. Clin Pharmacokinet. 26: 107-20. PMID: 8162655
- Clinical pharmacology of oxicams: new insights into the mechanisms of their dose-dependent toxicity. | Albengres, E., et al. 1993. Int J Tissue React. 15: 125-34. PMID: 8188448
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sudoxicam, 100 mg | sc-212968 | 100 mg | $400.00 |
