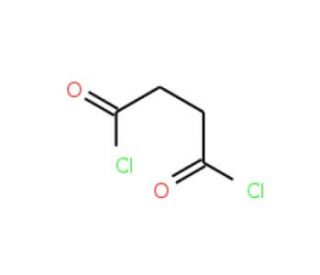
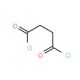
Succinyl chloride (CAS 543-20-4)
QUICK LINKS
Succinyl chloride is a chemical compound that functions as an acylating agent in organic synthesis. It is used to introduce the succinyl group into various organic compounds, particularly in the production of fine chemicals. The mechanism of action of succinyl chloride involves the reaction of the acyl chloride group with nucleophiles, such as amines or alcohols, to form amides or esters, respectively. Succinyl Chloride reaction in the modification of organic molecules to create new compounds with specific properties or functionalities. Succinyl chloride′s role in the acylation process is to facilitate the formation of covalent bonds between the succinyl group and the target molecules, enabling the synthesis of diverse chemical derivatives for development purposes. Its mechanism of action at the molecular level involves the transfer of the succinyl group to nucleophilic sites on the substrate, leading to the formation of the desired acylated products.
Succinyl chloride (CAS 543-20-4) References
- Resorbable and highly elastic block copolymers from 1,5-dioxepan-2-one and L-lactide with controlled tensile properties and hydrophilicity. | Ryner, M. and Albertsson, AC. 2002. Biomacromolecules. 3: 601-8. PMID: 12005533
- Highly active, regioselective, and enantioselective hydroformylation with Rh catalysts ligated by Bis-3,4-diazaphospholanes. | Clark, TP., et al. 2005. J Am Chem Soc. 127: 5040-2. PMID: 15810837
- Syntheses of new peptidic glycoclusters derived from beta-alanine: di- and trimerized glycoclusters and glycocluster-clusters. | Sato, K., et al. 2006. Carbohydr Res. 341: 836-45. PMID: 16530741
- Isosteric heats of sorption of 1-naphthol and phenol from aqueous solutions by beta-cyclodextrin polymers. | García-Zubiri, IX., et al. 2007. J Colloid Interface Sci. 307: 64-70. PMID: 17141797
- Synthesis, characterization and biological evaluation of succinate prodrugs of curcuminoids for colon cancer treatment. | Wichitnithad, W., et al. 2011. Molecules. 16: 1888-900. PMID: 21343891
- [10-Acyldithranol dimers; new derivatives of the antipsoriatic dithranol; synthesis, characterization and biochemical properties]. | Berset, JD. and Krebs, A. 1990. Pharm Acta Helv. 65: 155-64. PMID: 2371307
- Rapid separations by LC using ion-exchange media based on spongy monoliths. | Kubo, T., et al. 2013. J Sep Sci. 36: 2813-8. PMID: 23765523
- QuadraPure-supported palladium nanocatalysts for microwave-promoted Suzuki cross-coupling reaction under aerobic condition. | Liew, KH., et al. 2014. ScientificWorldJournal. 2014: 796196. PMID: 25054185
- Dual-pH Sensitive Charge-Reversal Polypeptide Micelles for Tumor-Triggered Targeting Uptake and Nuclear Drug Delivery. | Han, SS., et al. 2015. Small. 11: 2543-54. PMID: 25626995
- First use of grape waste-derived building blocks to yield antimicrobial materials. | Jelley, RE., et al. 2022. Food Chem. 370: 131025. PMID: 34509147
- [Anaphylactic shock induced by suxamethonium]. | Laxenaire, MC., et al. 1982. Ann Fr Anesth Reanim. 1: 29-36. PMID: 7137663
- Synthesis and physicochemical properties of the furan dicarboxylic acid, 3-carboxy-4-methyl-5-propyl-2-furanpropanoic acid, an inhibitor of plasma protein binding in uraemia. | Costigan, MG., et al. 1996. J Pharm Pharmacol. 48: 635-40. PMID: 8832500
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Succinyl chloride, 25 g | sc-258185 | 25 g | $23.00 |
