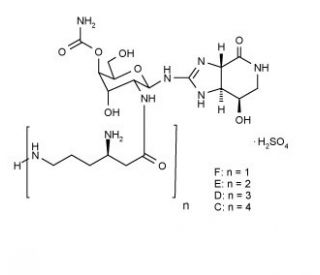
Streptothricin sulfate (CAS 96736-11-7)
See product citations (2)
QUICK LINKS
Streptothricin sulfate is a broad spectrum antibacterial compound with antiviral activity, composed of a streptolidine ring bound through carbon 1 of a 4-O-acetyl, N-poly-lysinylated glucosamine molecule. Candida albicans studies indicate that C. albicans is susceptible to Streptothricin sulfate. The compound is a mixture of Streptothricin D and Streptothricin F. Active against Gram-positive and Gram-negative bacteria, and some fungi.In scientific research, Streptothricin sulfate has demonstrated its utility across an array of contexts, notably in protein purification and gene expression analyses. This compound proves instrumental in extracting proteins from cellular lysates, as well as characterizing proteins sourced from cell cultures. Within gene expression inquiries, Streptothricin sulfate emerges as a means to affix fluorescent markers to proteins, thereby enabling the visualization and quantification of gene activity. The compound contributes to investigations into protein structure and function within cell cultures.
Streptothricin sulfate (CAS 96736-11-7) References
- Aminoglycoside-streptothricin resistance gene cluster aadE-sat4-aphA-3 disseminated among multiresistant isolates of Enterococcus faecium. | Werner, G., et al. 2001. Antimicrob Agents Chemother. 45: 3267-9. PMID: 11600397
- Enzymology of tubercle bacilli and other mycobacteria. V. Influence of streptomycin and other basic substances on the diamine oxidase of various bacteria. | OWEN, CA., et al. 1951. J Bacteriol. 62: 53-62. PMID: 14861159
- Cross-resistance studies with streptomycin, streptothricin, neomycin, and streptolin. | PAGANO, JF., et al. 1952. Proc Soc Exp Biol Med. 79: 359-63. PMID: 14920427
- CaNAT1, a heterologous dominant selectable marker for transformation of Candida albicans and other pathogenic Candida species. | Shen, J., et al. 2005. Infect Immun. 73: 1239-42. PMID: 15664973
- CRYSTALLINE REINECKATES OF STREPTOTHRICIN AND STREPTOMYCIN. | Fried, J. and Wintersteiner, O. 1945. Science. 101: 613-5. PMID: 17740751
- Streptomyces venezuelae, n. sp., the source of chloromycetin. | Ehrlich, J., et al. 1948. J Bacteriol. 56: 467-77. PMID: 18887825
- In Bacillus subtilis, the SatA (Formerly YyaR) Acetyltransferase Detoxifies Streptothricin via Lysine Acetylation. | Burckhardt, RM. and Escalante-Semerena, JC. 2017. Appl Environ Microbiol. 83: PMID: 28842538
- Chromatin Loop Formation Induced by a Subtelomeric Protosilencer Represses EPA Genes in Candida glabrata. | López-Fuentes, E., et al. 2018. Genetics. 210: 113-128. PMID: 30002080
- In vivo and in vitro inhibition of hepatitis viruses by streptothricin. | Stone, RL., et al. 1965. Ann N Y Acad Sci. 130: 355-64. PMID: 4285583
- Chromatographic fractionation of antibiotic 136 and characterization of its components. | Phillips, WF. and Ragheb, HS. 1965. J Chromatogr. 19: 147-59. PMID: 4285597
- Streptothricin biosynthesis is catalyzed by enzymes related to nonribosomal peptide bond formation. | Fernández-Moreno, MA., et al. 1997. J Bacteriol. 179: 6929-36. PMID: 9371436
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Streptothricin sulfate, 1 mg | sc-203284 | 1 mg | $38.00 | |||
Streptothricin sulfate, 5 mg | sc-203284A | 5 mg | $63.00 |
