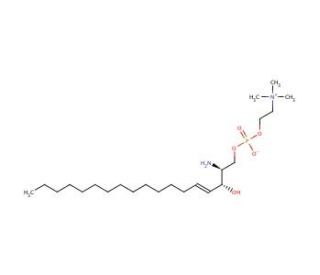
Sphingosylphosphorylcholine, D-erythro (CAS 1670-26-4)
See product citations (1)
QUICK LINKS
D-erythro-Sphingosine-1-phosphate (S1P) is a bioactive sphingolipid metabolite that acts as a potent signaling molecule involved in various cellular processes. S1P functions through specific G protein-coupled receptors (S1P receptors), known as S1P1-5, which are widely expressed in various tissues and cell types. Upon binding to its receptors, S1P regulates diverse cellular responses, including cell proliferation, migration, survival, and differentiation. In research, S1P has garnered significant attention for its roles in immunology, neuroscience, and cardiovascular biology. In immunology, S1P is recognized for its ability to modulate lymphocyte trafficking and immune cell activation, thereby influencing immune responses, inflammation, and autoimmunity. Furthermore, S1P signaling has implications in neurobiology, where it regulates neural development, synaptic plasticity, and neuroinflammation, suggesting its potential involvement in neurodegenerative diseases and neurological disorders. Additionally, S1P is implicated in cardiovascular physiology, contributing to vascular development, endothelial barrier function, and vascular tone regulation. Research efforts are underway to explain the precise mechanisms underlying S1P-mediated signaling pathways and its implications in various physiological and pathological conditions, offering insights into potential interventions targeting S1P signaling for the treatment of immune-related disorders, neurological diseases, and cardiovascular ailments.
Sphingosylphosphorylcholine, D-erythro (CAS 1670-26-4) References
- Effect of sphingosylphosphorylcholine on the single channel gating properties of the cardiac ryanodine receptor. | Uehara, A., et al. 1999. FEBS Lett. 460: 467-71. PMID: 10556518
- Methanolysis of sphingomyelin. Toward an epimerization-free methodology for the preparation of D-erythro-sphingosylphosphocholine. | Bittman, R. and Verbicky, CA. 2000. J Lipid Res. 41: 2089-93. PMID: 11108743
- Extra- and intracellular sphingosylphosphorylcholine promote spontaneous transmitter release from frog motor nerve endings. | Brailoiu, E. and Dun, NJ. 2003. Mol Pharmacol. 63: 1430-6. PMID: 12761354
- Role of MEK-ERK pathway in sphingosylphosphorylcholine-induced cell death in human adipose tissue-derived mesenchymal stem cells. | Jeon, ES., et al. 2005. Biochim Biophys Acta. 1734: 25-33. PMID: 15866480
- Sphingosylphosphorylcholine down-regulates filaggrin gene transcription through NOX5-based NADPH oxidase and cyclooxygenase-2 in human keratinocytes. | Choi, H., et al. 2010. Biochem Pharmacol. 80: 95-103. PMID: 20230798
- Structural role of mismatched C-C bonds in a series of d-erythro-sphingomyelins as studied by DSC and electron microscopy. | Kodama, M. and Kawasaki, Y. 2010. Chem Phys Lipids. 163: 514-23. PMID: 20307518
- Sphingosylphosphocholine, a signaling molecule which accumulates in Niemann-Pick disease type A, stimulates DNA-binding activity of the transcription activator protein AP-1. | Berger, A., et al. 1995. Proc Natl Acad Sci U S A. 92: 5885-9. PMID: 7597047
- Signaling pathways for sphingosylphosphorylcholine-mediated mitogenesis in Swiss 3T3 fibroblasts. | Desai, NN., et al. 1993. J Cell Biol. 121: 1385-95. PMID: 8389770
- Sphingosine metabolism induces Ca2+ oscillations in rat pancreatic acinar cells. | Yule, DI., et al. 1993. J Biol Chem. 268: 12353-8. PMID: 8509375
- Preparation of a naturally occurring D-erythro-(2S, 3R)-sphingosylphosphocholine using Shewanella alga NS-589. | Sueyoshi, N., et al. 1997. J Lipid Res. 38: 1923-7. PMID: 9323602
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sphingosylphosphorylcholine, D-erythro, 10 mg | sc-201384 | 10 mg | $408.00 |
