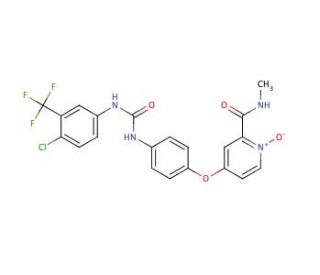
Molecular structure of Sorafenib N-Oxide, CAS Number: 583840-03-3
Sorafenib N-Oxide (CAS 583840-03-3)
Alternate Names:
Sorafenib N-Oxide is also known as BAY 67-3472.
Application:
Sorafenib N-Oxide is an active metabolite of Sorafenib, a potent Raf kinase, FLT3, and acute myeloid leukemia cell inhibitor.
CAS Number:
583840-03-3
Molecular Weight:
480.82
Molecular Formula:
C21H16ClF3N4O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Sorafenib N-Oxide is a metabolite of Sorafenib, a potent Raf kinase inhibitor. In addition to Raf-1, B-RAF, and receptor tyrosine kinases, Sorafenib N-Oxide also inhibits FLT3 that contains the internal tandem duplication mutation and inhibits proliferation of MV4-11 acute myeloid leukemia (AML) cells expressing FLT3-ITD. Sorafenib N-Oxide is selective for AML cell lines containing FLT3-ITD over lines containing wild FLT3. Sorafenib N-Oxide can function as a linear-mixed inhibitor of CYP3A4. Sorafenib N-oxide acts as a linear-mixed inhibitor of the cytochrome P450 (CYP) isoform CYP3A4 with a Ki value of 15 µM in human liver microsomes.
Sorafenib N-Oxide (CAS 583840-03-3) References
- Quantitation of sorafenib and its active metabolite sorafenib N-oxide in human plasma by liquid chromatography-tandem mass spectrometry. | Li, L., et al. 2010. J Chromatogr B Analyt Technol Biomed Life Sci. 878: 3033-8. PMID: 20870468
- A quantitative HPLC-UV method for determination of serum sorafenib and sorafenib N-oxide and its application in hepatocarcinoma patients. | Shimada, M., et al. 2014. Tohoku J Exp Med. 233: 103-12. PMID: 24872323
- The Influence of Paracetamol on the Penetration of Sorafenib and Sorafenib N-Oxide Through the Blood-Brain Barrier in Rats. | Karbownik, A., et al. 2020. Eur J Drug Metab Pharmacokinet. 45: 801-808. PMID: 32776310
- Baicalin Enhanced Oral Bioavailability of Sorafenib in Rats by Inducing Intestine Absorption. | Wei, J., et al. 2021. Front Pharmacol. 12: 761763. PMID: 34819863
- Pharmacokinetic Drug Interaction Study of Sorafenib and Morphine in Rats. | Karbownik, A., et al. 2021. Pharmaceutics. 13: PMID: 34959453
- The Role of Uptake and Efflux Transporters in the Disposition of Glucuronide and Sulfate Conjugates. | Järvinen, E., et al. 2021. Front Pharmacol. 12: 802539. PMID: 35095509
- Pharmacokinetic Exposures Associated With Oral Administration of Sorafenib in Dogs With Spontaneous Tumors. | Cawley, JR., et al. 2022. Front Vet Sci. 9: 888483. PMID: 35664857
- Hesperetin mitigates sorafenib-induced cardiotoxicity in mice through inhibition of the TLR4/NLRP3 signaling pathway. | Zaafar, D., et al. 2022. PLoS One. 17: e0271631. PMID: 35944026
- Pharmacokinetic Interactions between Canagliflozin and Sorafenib or Lenvatinib in Rats. | Cui, Y., et al. 2022. Molecules. 27: PMID: 36080187
- A high throughput method for Monitoring of Sorafenib, regorafenib, cabozantinib and their metabolites with UPLC-MS/MS in rat plasma. | Gu, EM., et al. 2022. Front Pharmacol. 13: 955263. PMID: 36160432
- Inhibitory effects of Thai herbal extracts on the cytochrome P450 3A-mediated the metabolism of gefitinib, lapatinib and sorafenib. | Rodseeda, C., et al. 2022. Toxicol Rep. 9: 1846-1852. PMID: 36518483
- Efficacy and safety of lenvatinib versus sorafenib in first-line treatment of advanced hepatocellular carcinoma: A meta-analysis. | Luo, J., et al. 2022. Front Oncol. 12: 1010726. PMID: 36620586
- The Role of Organic Cation Transporters in the Pharmacokinetics, Pharmacodynamics and Drug-Drug Interactions of Tyrosine Kinase Inhibitors. | Xiu, F., et al. 2023. Int J Mol Sci. 24: PMID: 36768423
Inhibitor of:
Raf.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sorafenib N-Oxide, 1 mg | sc-208399A | 1 mg | $203.00 | |||
Sorafenib N-Oxide, 5 mg | sc-208399 | 5 mg | $490.00 | |||
Sorafenib N-Oxide, 25 mg | sc-208399B | 25 mg | $1775.00 | |||
Sorafenib N-Oxide, 50 mg | sc-208399C | 50 mg | $3060.00 |
