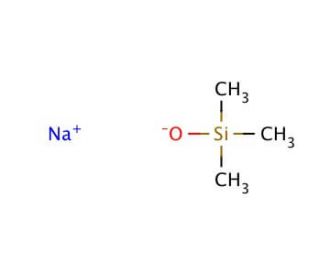
Sodium trimethylsilanolate (CAS 18027-10-6)
QUICK LINKS
Sodium trimethylsilanolate is a chemical compound that functions as a strong base in various experimental. Its mode of action involves the deprotonation of acidic compounds, leading to the formation of silanolates. This process facilitates the synthesis of organosilicon compounds and the modification of surfaces through silanization reactions. Sodium trimethylsilanolate can act as a catalyst in the formation of silicon-containing polymers, contributing to the development of new materials with unique properties. Its ability to promote condensation reactions makes it useful in the preparation of silicon-based nanoparticles and thin films. Sodium trimethylsilanolate can participate in the functionalization of organic molecules, enabling the creation of novel chemical structures for development purposes.
Sodium trimethylsilanolate (CAS 18027-10-6) References
- Automated carboxy-terminal sequence analysis of peptides. | Bailey, JM., et al. 1992. Protein Sci. 1: 68-80. PMID: 1304884
- Automated carboxy-terminal sequence analysis of peptides and proteins using diphenyl phosphoroisothiocyanatidate. | Bailey, JM., et al. 1992. Protein Sci. 1: 1622-33. PMID: 1304893
- Nitric oxide-releasing xerogels synthesized from N-diazeniumdiolate-modified silane precursors. | Storm, WL. and Schoenfisch, MH. 2013. ACS Appl Mater Interfaces. 5: 4904-12. PMID: 23651116
- N-acetylgalactosamino dendrons as clearing agents to enhance liver targeting of model antibody-fusion protein. | Yoo, B., et al. 2013. Bioconjug Chem. 24: 2088-103. PMID: 24147780
- Mild and chemoselective lactone ring-opening with (TMS)ONa. Mechanistic studies and application to sweroside derivatives. | Lemoine, H., et al. 2014. J Org Chem. 79: 4358-66. PMID: 24738692
- Atomic layer deposition of sodium and potassium oxides: evaluation of precursors and deposition of thin films. | Østreng, E., et al. 2014. Dalton Trans. 43: 16666-72. PMID: 25265332
- Copper-Catalyzed Alkylation of Nitroalkanes with α-Bromonitriles: Synthesis of β-Cyanonitroalkanes. | Shimkin, KW., et al. 2016. Org Lett. 18: 988-91. PMID: 26866576
- Metal-Metal Bonding in Uranium-Group 10 Complexes. | Hlina, JA., et al. 2016. J Am Chem Soc. 138: 3333-45. PMID: 26942560
- Asymmetric Hydroarylation of Vinylarenes Using a Synergistic Combination of CuH and Pd Catalysis. | Friis, SD., et al. 2016. J Am Chem Soc. 138: 8372-5. PMID: 27346525
- Trifluoromethylation of Secondary Nitroalkanes. | Gietter-Burch, AAS., et al. 2017. Org Lett. 19: 2957-2960. PMID: 28535057
- Sodium silylsilanolate as a precursor of silylcopper species. | Yamagishi, H., et al. 2022. Chem Sci. 13: 4334-4340. PMID: 35509465
- Pseudo-mono-axial ligand fields that support high energy barriers in triangular dodecahedral Dy(iii) single-ion magnets. | Zhang, B., et al. 2022. Chem Sci. 13: 13231-13240. PMID: 36425507
- Automated carboxy-terminal sequence analysis of polypeptides containing C-terminal proline. | Bailey, JM., et al. 1995. Anal Biochem. 224: 588-96. PMID: 7733462
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sodium trimethylsilanolate, 25 g | sc-229324 | 25 g | $98.00 | |||
Sodium trimethylsilanolate, 100 g | sc-229324A | 100 g | $417.00 |
