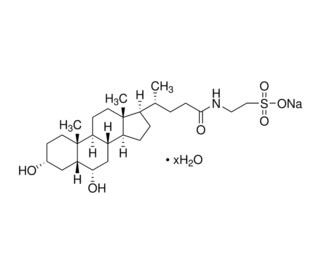
Sodium taurohyodeoxycholate (CAS 38411-85-7)
QUICK LINKS
Sodium taurohyodeoxycholate (STDC) represents a bile acid salt derived from the combination of taurine and hyodeoxycholic acid. This compound, possessing detergent-like properties, finds extensive application in laboratory experiments for the solubilization of membrane proteins. Researchers favor Sodium taurohyodeoxycholate due to its remarkable solubility, minimal toxicity, and reasonable cost. Sodium taurohyodeoxycholate has played a pivotal role across diverse scientific research applications, enabling the exploration of membrane protein structure, function, and their interactions with other proteins and small molecules. It has also contributed to our understanding of cell membrane structure, function, and interactions with other compounds. The mechanism of action for sodium taurohyodeoxycholate involves the disruption of the cell membrane, thereby facilitating the solubilization of membrane proteins. This disruption occurs through the binding of sodium taurohyodeoxycholate to the membrane lipids, inducing increased fluidity and allowing for the solubilization of membrane proteins.
Sodium taurohyodeoxycholate (CAS 38411-85-7) References
- Spontaneous and bile salt stimulated bile secretion in the Adelie penguin (Pygoscelis adeliae). | Andrews, CJ. 1978. Q J Exp Physiol Cogn Med Sci. 63: 23-37. PMID: 244360
- Identification of Dose-Dependent DNA Damage and Repair Responses From Subchronic Exposure to 1,4-Dioxane in Mice Using a Systems Analysis Approach. | Charkoftaki, G., et al. 2021. Toxicol Sci. 183: 338-351. PMID: 33693819
- Development of a Highly Sensitive Ultra-High-Performance Liquid Chromatography Coupled to Electrospray Ionization Tandem Mass Spectrometry Quantitation Method for Fecal Bile Acids and Application on Crohn's Disease Studies. | Reiter, S., et al. 2021. J Agric Food Chem. 69: 5238-5251. PMID: 33890469
- Targeted LC-MS/MS Profiling of Bile Acids in Various Animal Tissues. | Reiter, S., et al. 2021. J Agric Food Chem. 69: 10572-10580. PMID: 34490775
- Liver metabolomics identifies bile acid profile changes at early stages of alcoholic liver disease in mice. | Charkoftaki, G., et al. 2022. Chem Biol Interact. 360: 109931. PMID: 35429548
- Bile acids serve as endogenous antagonists of the Leukemia inhibitory factor (LIF) receptor in oncogenesis. | Di Giorgio, C., et al. 2024. Biochem Pharmacol. 223: 116134. PMID: 38494064
- Bile acid sulfonates alter cholesterol gallstone incidence in hamsters. | Cohen, BI., et al. 1993. Hepatology. 17: 103-10. PMID: 8423031
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sodium taurohyodeoxycholate, 250 mg | sc-281149 | 250 mg | $214.00 | |||
Sodium taurohyodeoxycholate, 1 g | sc-281149A | 1 g | $632.00 |
