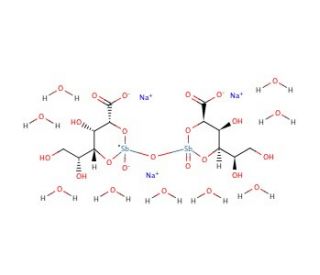
Molecular structure of Sodium stibogluconate, CAS Number: 16037-91-5
Sodium stibogluconate (CAS 16037-91-5)
See product citations (6)
Alternate Names:
2,4:2′,4′-O-(Oxy-distibylidyne)bis[D-gluconic acid] Sb,Sb-dioxide trisodium salt
Application:
Sodium stibogluconate is a robust inhibitor of PTPases, including PTPase1 (SHP-1), SHP-2, and PTP1B
CAS Number:
16037-91-5
Purity:
≥90%
Molecular Weight:
907.86
Molecular Formula:
C12H17O17Sb2•3Na•9H2O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Sodium stibogluconate augments cytokine response and is a robust inhibitor of PTPases, including PTPase1 (SHP-1), SHP-2, and PTP1B. SHP-1 activity has been reported to be inhibited by Sodium stibogluconate at a lower concentration than PTP1B and SHP-2. Studies indicate that Sodium stibogluconate can form a complex with the PTPase and in Baf3 cells increases IL-3-induced proliferation. Sodium stibogluconate has been used in multiple studies investigating leishmaniasis.
Sodium stibogluconate (CAS 16037-91-5) References
- Sodium stibogluconate is a potent inhibitor of protein tyrosine phosphatases and augments cytokine responses in hemopoietic cell lines. | Pathak, MK. and Yi, T. 2001. J Immunol. 167: 3391-7. PMID: 11544330
- A randomized, double-blind study of the efficacy of a 10- or 20-day course of sodium stibogluconate for treatment of cutaneous leishmaniasis in United States military personnel. | Wortmann, G., et al. 2002. Clin Infect Dis. 35: 261-7. PMID: 12115091
- Recommendations for treating leishmaniasis with sodium stibogluconate (Pentostam) and review of pertinent clinical studies. | Herwaldt, BL. and Berman, JD. 1992. Am J Trop Med Hyg. 46: 296-306. PMID: 1313656
- Sodium stibogluconate cardiotoxicity and safety of generics. | Rijal, S., et al. 2003. Trans R Soc Trop Med Hyg. 97: 597-8. PMID: 15307436
- Successful use of miltefosine and sodium stibogluconate, in combination, for the treatment of an HIV-positive patient with visceral leishmaniasis: a case report and brief review of the literature. | Collini, P., et al. 2009. Ann Trop Med Parasitol. 103: 455-9. PMID: 19583915
- Intralesional sodium stibogluconate alone or its combination with either intramuscular sodium stibogluconate or oral ketoconazole in the treatment of localized cutaneous leishmaniasis: a comparative study. | El-Sayed, M. and Anwar, AE. 2010. J Eur Acad Dermatol Venereol. 24: 335-40. PMID: 19744259
- A retrospective study of intravenous sodium stibogluconate alone and in combinations with allopurinol, rifampicin, and an immunomodulator in the treatment of Indian post-kala-azar dermal leishmaniasis. | Ramesh, V., et al. 2010. Indian J Dermatol Venereol Leprol. 76: 138-44. PMID: 20228542
- Sodium stibogluconate-associated acute interstitial nephritis in a patient treated for visceral leishmaniasis. | Vikrant, S., et al. 2015. Saudi J Kidney Dis Transpl. 26: 757-60. PMID: 26178551
- Sodium stibogluconate and paromomycin for treating visceral leishmaniasis under routine conditions in eastern Sudan. | Atia, AM., et al. 2015. Trop Med Int Health. 20: 1674-84. PMID: 26427033
- Sodium stibogluconate loaded nano-deformable liposomes for topical treatment of leishmaniasis: macrophage as a target cell. | Dar, MJ., et al. 2018. Drug Deliv. 25: 1595-1606. PMID: 30105918
- Intralesional sodium stibogluconate under inhaled anesthesia for the treatment of cutaneous leishmaniasis in children: A retrospective cohort. | Renert-Yuval, Y., et al. 2019. J Am Acad Dermatol. 81: 1013-1015. PMID: 30731176
- Treatment failure to sodium stibogluconate in cutaneous leishmaniasis: A challenge to infection control and disease elimination. | Silva, H., et al. 2021. PLoS One. 16: e0259009. PMID: 34679130
Inhibitor of:
DEP-1, DUSP, Hemoglobin μ, HLA-E, KIR2DL2, KLRH1, Pez, PTPλ, PTPDC1, PTPN20, PTPN21, PTPRCAP, SH-PTP1, SH-PTP2, and SHP.Activator of:
CD45RC, DUSP26, H2-Adβ, PTPε, SLA, and TC-PTP.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sodium stibogluconate, 1 g | sc-202815 | 1 g | $188.00 |
