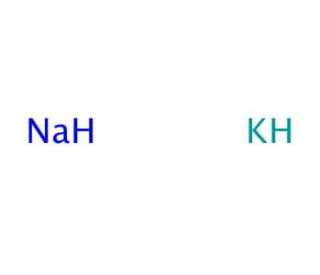
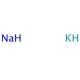
Sodium-potassium alloy (CAS 11135-81-2)
Available in US only.
QUICK LINKS
Sodium-potassium alloy, commonly referred to as NaK, is an intriguing metal alloy consisting of sodium (Na) and potassium (K), both alkali metals. This alloy is notable for being liquid at room temperature, primarily due to the lowering of the melting point below that of either of its constituent metals when combined. NaK is typically composed in various ratios, although a common formulation is 40% sodium and 60% potassium by weight, which presents a lower melting point around -12.6 degrees Celsius. This unique physical property makes NaK particularly useful in a range of scientific and industrial applications where heat transfer is essential. In research, NaK has been extensively used as a coolant in experimental nuclear reactors and other high-temperature engineering processes due to its excellent thermal conductivity and wide liquid phase range. Its ability to efficiently transfer heat at relatively low temperatures compared to pure metals makes it ideal for maintaining controlled temperatures in systems requiring precise thermal management. Additionally, NaK is utilized in various specialized chemical syntheses and reactions where the reactivity of alkali metals is needed without the hazards of handling them in their pure, more reactive forms. For instance, in synthetic chemistry, it facilitates reactions in organic synthesis where the dissolution or dispersion of metals in solvents is necessary, but the high reactivity of pure sodium or potassium could be problematic. This blend of unique physical and chemical properties underscores the versatility and utility of sodium-potassium alloy in both research and industrial contexts.
Sodium-potassium alloy (CAS 11135-81-2) References
- μ-Methyl-ene-bis-[di-bromido(diethyl ether-κO)aluminium(III)]: crystal structure and chemical exchange in solution. | Butcher, RJ. and Purdy, AP. 2021. Acta Crystallogr E Crystallogr Commun. 77: 647-652. PMID: 34164145
- Flow and Heat Transfer Performances of Liquid Metal Based Microchannel Heat Sinks under High Temperature Conditions. | Wu, T., et al. 2022. Micromachines (Basel). 13: PMID: 35056260
- Radical Covalent Organic Frameworks Associated with Liquid Na-K toward Dendrite-Free Alkali Metal Anodes. | Wang, J., et al. 2022. Adv Sci (Weinh). 9: e2203058. PMID: 35861409
- Selective synthesis of germasila-adamantanes through germanium-silicon shift processes. | Kühn, S., et al. 2023. Chem Sci. 14: 8956-8961. PMID: 37621423
- Cobalt/Nitrogen Co-Doped Carbon Materials Enhance the Reaction Rate of Sodium-Potassium Alloy Electrodes. | Luo, Y., et al. 2024. Small. 20: e2304981. PMID: 37672807
- Synthesis and Thermoreversible Gelation of Coil-Rod Copolymers with a Dendritic Polyethylene Core and Multiple Helical Poly(γ-benzyl-L-glutamate) Arms. | Lu, Y., et al. 2023. Polymers (Basel). 15: PMID: 38006076
- A Novel Sodium-Potassium Anode Supported by Fluorinated Aluminum Foam. | Lou, J., et al. 2023. Materials (Basel). 16: PMID: 38068013
- Synthesis and Spectroscopic Characterization of Selected Water-Soluble Ligands Based on 1,10-Phenanthroline Core. | Nycz, JE., et al. 2024. Molecules. 29: PMID: 38542977
- Position-Dependent Segmental Relaxation in Bottlebrush Polymers. | Bichler, KJ., et al. 2024. Macromolecules. 57: 4729-4736. PMID: 38827960
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sodium-potassium alloy, 1 g | sc-281150 | 1 g | $45.00 | |||
| US: Only available in the US | ||||||
