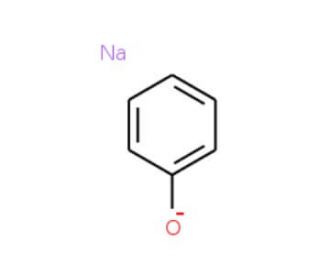
Molecular structure of Sodium phenoxide, CAS Number: 139-02-6
Sodium phenoxide (CAS 139-02-6)
Alternate Names:
Phenolate sodium
CAS Number:
139-02-6
Purity:
≥97%
Molecular Weight:
116.09
Molecular Formula:
C6H5ONa
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Sodium phenoxide functions as a strong base in organic synthesis. It is used as a nucleophile in various reactions, such as nucleophilic aromatic substitution and the Williamson ether synthesis. In these processes, sodium phenoxide acts by donating an electron pair to an electrophilic substrate, leading to the formation of new carbon-carbon or carbon-oxygen bonds. The mechanism of action involves the deprotonation of the phenol group, resulting in the formation of the phenoxide ion, which is highly reactive towards electrophiles. Its ability to facilitate the formation of new chemical bonds at the molecular level makes it useful in the development of novel compounds and materials.
Sodium phenoxide (CAS 139-02-6) References
- Chemistry of Bis(aryloxo)palladium(II) Complexes with N-Donor Ligands: Structural Features of the Palladium-to-Oxygen Bond and Formation of O-H.O Bonds. | Kapteijn, GM., et al. 1996. Inorg Chem. 35: 526-533. PMID: 11666240
- Azidophosphazenes as Functionalized Intermediates. | Allcock, HR., et al. 1999. Inorg Chem. 38: 5535-5544. PMID: 11671281
- Studies of chemical selectivity of hapten, reactivity, and skin sensitization potency. 3. Synthesis and studies on the reactivity toward model nucleophiles of the 13C-labeled skin sensitizers, 5-chloro-2-methylisothiazol-3-one (MCI) and 2-methylisothiazol-3-one (MI). | Alvarez-Sánchez, R., et al. 2003. Chem Res Toxicol. 16: 627-36. PMID: 12755592
- Chloromethyl chlorosulfate: a new, catalytic method of preparation and reactions with some nucleophiles. | Power, NP., et al. 2004. Org Biomol Chem. 2: 1554-62. PMID: 15136814
- Palladium-catalyzed aminocarbonylation of aryl chlorides at atmospheric pressure: the dual role of sodium phenoxide. | Martinelli, JR., et al. 2007. Angew Chem Int Ed Engl. 46: 8460-3. PMID: 17902084
- Heterobimetallic lanthanide/sodium phenoxides: efficient catalysts for amidation of aldehydes with amines. | Li, J., et al. 2009. J Org Chem. 74: 2575-7. PMID: 19209872
- Reversing the stereoselectivity of a palladium-catalyzed O-glycosylation through an inner-sphere or outer-sphere pathway. | Xiang, S., et al. 2015. Angew Chem Int Ed Engl. 54: 604-7. PMID: 25366278
- Characterization of char from slow pyrolysis of sewage sludge. | Xu, WY. and Wu, D. 2016. Water Sci Technol. 73: 2370-8. PMID: 27191557
- New Synthesis of Optically Active O-Aryl O-Ethyl Phenylphosphonothionates. | Yoshikawa, H. 1999. Biosci Biotechnol Biochem. 63: 424-6. PMID: 27393069
- Preparation of Poly(bis(phenoxy)phosphazene) and 31P NMR Analysis of Its Structural Defects under Various Synthesis Conditions. | Zhang, S., et al. 2016. J Phys Chem B. 120: 11307-11316. PMID: 27726395
- A Teaching Experiment to Elucidate a Cation-π Effect in an Alkyne Cycloaddition Reaction and Illustrate Hypothesis Driven Design of Experiments. | St Germain, EJ., et al. 2017. J Chem Educ. 94: 240-243. PMID: 29200498
- Realizing room temperature catalytic hydrogenation of sodium phenoxide by Ru/TiO2 for hydrogen storage. | Tan, KC., et al. 2023. Chem Commun (Camb). 59: 4177-4180. PMID: 36942825
- Structure of 1, 6-bis (p-chlorophenyl)-3, 4-diacetyl-1, 5-hexaazadiene: a compound with a highly electrophilic N-acetyl group | Mackay, D., McIntyre, D. D., & Taylor, N. J. 1982. The Journal of Organic Chemistry. 47(3): 532-535.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sodium phenoxide, 50 g | sc-272494 | 50 g | $112.00 |
