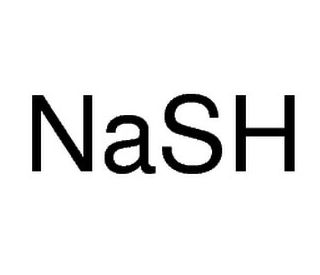
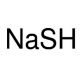
Sodium hydrosulfide (CAS 207683-19-0)
See product citations (1)
QUICK LINKS
Sodium hydrosulfide is an important sulfur compound primarily used as a reducing agent and a sulfur source in various chemical reactions. It is highly soluble in water, forming an alkaline solution that is widely utilized in the leather industry for tanning processes. Additionally, it is employed in the production of dyes, flotation agents, and other chemicals. In scientific research, sodium hydrosulfide finds applications in studies involving sulfur chemistry, synthesis of organic compounds, and as a precursor for generating hydrogen sulfide (H2S) gas. The mechanism of action of sodium hydrosulfide primarily revolves around its ability to release hydrogen sulfide (H2S) when dissolved in water. H2S is a gas that can act as a reducing agent and participate in various chemical reactions. Sodium hydrosulfide provides a convenient and controllable source of H2S, allowing researchers to study its effects on biological systems, signaling pathways, and physiological processes. It is an H2S donor used in cellular and whole animal experimental systems.
Sodium hydrosulfide (CAS 207683-19-0) References
- Differentiated astrocytes acquire sensitivity to hydrogen sulfide that is diminished by the transformation into reactive astrocytes. | Tsugane, M., et al. 2007. Antioxid Redox Signal. 9: 257-69. PMID: 17115938
- Sodium hydrosulfide alleviates pulmonary artery collagen remodeling in rats with high pulmonary blood flow. | Li, X., et al. 2008. Heart Vessels. 23: 409-19. PMID: 19037589
- Initial characterization of hydrogen sulfide effects in female sexual function. | Srilatha, B., et al. 2009. J Sex Med. 6: 1875-84. PMID: 19453900
- A general and straightforward method for the synthesis of 2-trifluoromethylbenzothiazoles. | Zhu, J., et al. 2010. Org Lett. 12: 2434-6. PMID: 20402511
- Effect of hypoxia in mice mesenteric arteries surrounded by adipose tissue. | Maenhaut, N. and Van de Voorde, J. 2011. Acta Physiol (Oxf). 203: 235-44. PMID: 21362151
- Endogenous hydrogen sulfide insufficiency as a predictor of sexual dysfunction in aging rats. | Srilatha, B., et al. 2012. Aging Male. 15: 153-8. PMID: 22455401
- Depolarizing actions of hydrogen sulfide on hypothalamic paraventricular nucleus neurons. | Khademullah, CS. and Ferguson, AV. 2013. PLoS One. 8: e64495. PMID: 23691233
- Hydrogen sulfide in posthemorrhagic shock mesenteric lymph drainage alleviates kidney injury in rats. | Han, B., et al. 2015. Braz J Med Biol Res. 48: 622-8. PMID: 25945746
- Hydrogen Sulfide Facilitates Vaginal Lubrication by Activation of Epithelial ATP-Sensitive K(+) Channels and Cystic Fibrosis Transmembrane Conductance Regulator. | Sun, Q., et al. 2016. J Sex Med. 13: 798-807. PMID: 27114193
- Cerebroprotective effects of hydrogen sulfide in homocysteine-induced neurovascular permeability: Involvement of oxidative stress, arginase, and matrix metalloproteinase-9. | Nath, N., et al. 2019. J Cell Physiol. 234: 3007-3019. PMID: 30206943
- Hepatoprotective effect of sodium hydrosulfide on hepatic encephalopathy in rats. | Kwon, KW., et al. 2019. Korean J Physiol Pharmacol. 23: 263-270. PMID: 31297010
- Hydrogen sulfide (H2S) and nitric oxide (NO) alleviate cobalt toxicity in wheat (Triticum aestivum L.) by modulating photosynthesis, chloroplastic redox and antioxidant capacity. | Ozfidan-Konakci, C., et al. 2020. J Hazard Mater. 388: 122061. PMID: 31954305
- Nanofibrous Membrane Dressings Loaded With Sodium Hydrogen Sulfide/Endothelial Progenitor Cells Promote Wound Healing. | Lian, J., et al. 2021. Front Bioeng Biotechnol. 9: 657549. PMID: 34422776
- Protective effect of H2S on LPS‑induced AKI by promoting autophagy. | Li, T., et al. 2022. Mol Med Rep. 25: PMID: 35059738
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sodium hydrosulfide, 5 g | sc-229304 | 5 g | $53.00 | |||
Sodium hydrosulfide, 100 g | sc-229304A | 100 g | $63.00 | |||
Sodium hydrosulfide, 1 kg | sc-229304B | 1 kg | $94.00 | |||
Sodium hydrosulfide, 3 kg | sc-229304C | 3 kg | $119.00 |
