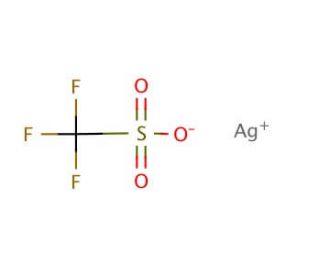
Molecular structure of Silver trifluoromethanesulfonate, CAS Number: 2923-28-6
Silver trifluoromethanesulfonate (CAS 2923-28-6)
Alternate Names:
Silver triflate; Trifluoromethanesulfonic acid silver salt
CAS Number:
2923-28-6
Purity:
≥98%
Molecular Weight:
256.94
Molecular Formula:
CF3SO3Ag
Supplemental Information:
This is as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Silver trifluoromethanesulfonate from secondary phosphates and thiophosphates. Using this reagent the etherification of alcohols with primary alkyl halides can be accomplished under mild conditions. Used to generate cationic rhodium catalysts from chlororhodium complexes for the hydrophosphination of acetylenes. Additionally, employed as a catalyst for the preparation of silyl ethers by hydrosilylation of aldehydes.
Silver trifluoromethanesulfonate (CAS 2923-28-6) References
- Brominations of steroidal hormone having alpha,beta-unsaturated ketone, 17-O-acetyltestosterone, in the presence of silver triflate. | Shimizu, T., et al. 2001. Chem Pharm Bull (Tokyo). 49: 23-8. PMID: 11201220
- Iodine monochloride/silver trifluoromethanesulfonate (ICI/AgOTf) as a convenient promoter system for O-glycoside synthesis. | Ercegovic, T., et al. 2001. Org Lett. 3: 913-5. PMID: 11263914
- Study of interhalogens/silver trifluoromethanesulfonate as promoter systems for high-yielding sialylations. | Meijer, A. and Ellervik, U. 2002. J Org Chem. 67: 7407-12. PMID: 12375971
- A stable, commercially available sulfenyl chloride for the activation of thioglycosides in conjunction with silver trifluoromethanesulfonate. | Crich, D., et al. 2008. Carbohydr Res. 343: 1858-62. PMID: 18374318
- Cation-induced pi-stacking. | Bosch, E., et al. 2008. J Org Chem. 73: 3931-4. PMID: 18407690
- Syntheses, structures and spectroscopy of uni- and bi-dentate nitrogen base complexes of silver(I) trifluoromethanesulfonate. | Di Nicola, C., et al. 2010. Dalton Trans. 39: 908-22. PMID: 20066236
- A direct and mild formylation method for substituted benzenes utilizing dichloromethyl methyl ether-silver trifluoromethanesulfonate. | Ohsawa, K., et al. 2013. J Org Chem. 78: 3438-44. PMID: 23477294
- A Convergent Synthesis of Homogeneous Reducible Polypeptides. | Ericson, MD. and Rice, KG. 2013. Tetrahedron Lett. 54: 4746-4748. PMID: 24493905
- Silver Trifluoromethanesulfonate-Catalyzed Annulation of Propargylic Alcohols with 3-Methyleneisoindolin-1-one. | Li, XS., et al. 2020. J Org Chem. 85: 2626-2634. PMID: 31880453
- Synthesis, Spectroscopy, Electrochemistry and DFT of Electron-Rich Ferrocenylsubphthalocyanines. | Swarts, PJ. and Conradie, J. 2020. Molecules. 25: PMID: 32492953
- Characterization of the O-Glycoproteome of Tannerella forsythia. | Veith, PD., et al. 2021. mSphere. 6: e0064921. PMID: 34523981
- Ionic Liquids Effect on the Stability of 17-Electron Cation Product of the Electrochemical Oxidation of Cymantrene. | Torriero, AAJ. 2022. Molecules. 27: PMID: 36364253
- Catalytic aza-Nazarov cyclization reactions to access α-methylene-γ-lactam heterocycles. | Yagci, BB., et al. 2023. Beilstein J Org Chem. 19: 66-77. PMID: 36741815
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Silver trifluoromethanesulfonate, 1 g | sc-255615 | 1 g | $24.00 | |||
Silver trifluoromethanesulfonate, 10 g | sc-255615A | 10 g | $84.00 | |||
Silver trifluoromethanesulfonate, 100 g | sc-255615B | 100 g | $656.00 | |||
Silver trifluoromethanesulfonate, 1 kg | sc-255615C | 1 kg | $4162.00 |
