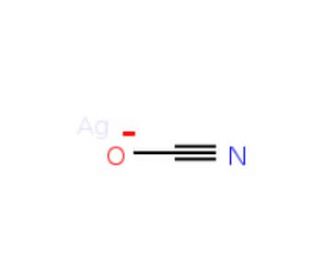
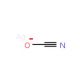
Silver cyanate (CAS 3315-16-0)
QUICK LINKS
Silver cyanate, consisting of silver and cyanate ions, is a inorganic compound employed in various scientific and industrial endeavors. It serves as a fundamental chemical compound with diverse applications. Silver cyanate finds utility in organic compound synthesis, functioning as an oxidizing agent and catalyst in organic reactions. In scientific research, silver cyanate exhibits a wide range of applications. It serves as useful in organic compound synthesis, as an oxidizing agent, and as a catalyst for organic reactions. Although the exact mechanism of action of silver cyanate remains incompletely understood, the current understanding posits that the silver ion within the silver cyanate molecule functions as a Lewis acid, while the cyanate ion acts as a Lewis base. The silver ion is believed to be responsible for the compound′s oxidizing properties, whereas the cyanate ion is thought to contribute to its catalytic properties.
Silver cyanate (CAS 3315-16-0) References
- Syntheses of carbocyclic uracil polyoxin C analogs: application of Pd(0)/InI-allylation of 4-acetoxy-2-azetidinone. | Cesario, C. and Miller, MJ. 2009. J Org Chem. 74: 5730-3. PMID: 19514728
- Palladium-catalyzed cross-coupling of aryl chlorides and triflates with sodium cyanate: a practical synthesis of unsymmetrical ureas. | Vinogradova, EV., et al. 2012. J Am Chem Soc. 134: 11132-5. PMID: 22716197
- An efficient and practical radiosynthesis of [11C]temozolomide. | Moseley, CK., et al. 2012. Org Lett. 14: 5872-5. PMID: 23151019
- Synthesis of a North-methanocarba-thymidine (N-MCT) analog. | Thompson, A. and Marquez, VE. 2012. Curr Protoc Nucleic Acid Chem. Chapter 1: Unit1.29. PMID: 23255201
- Radiosynthesis and reactivity of N-[11C]methyl carbamoylimidazole. | Kadirvel, M., et al. 2018. J Radioanal Nucl Chem. 317: 977-984. PMID: 30100651
- Defining the Scope of the Acid-Catalyzed Glycosidation of Glycosyl Bromides. | Singh, Y. and Demchenko, AV. 2020. Chemistry. 26: 1042-1051. PMID: 31614042
- Synthesis, antiviral, cytotoxic and cytostatic evaluation of N 1-(phosphonoalkyl)uracil derivatives. | Rygielska-Tokarska, D., et al. 2016. Monatsh Chem. 147: 1081-1090. PMID: 32214481
- Synthesis and Evaluation of Artificial Nucleic Acid Bearing an Oxanorbornane Scaffold. | Komine, H., et al. 2020. Molecules. 25: PMID: 32283778
- Perspectives from nearly five decades of total synthesis of natural products and their analogues for biology and medicine. | Nicolaou, KC. and Rigol, S. 2020. Nat Prod Rep. 37: 1404-1435. PMID: 32319494
- Chemical Bonding: The Journey from Miniature Hooks to Density Functional Theory. | Constable, EC. and Housecroft, CE. 2020. Molecules. 25: PMID: 32516906
- Enantiocontrolled Preparation of ϒ-Substituted Cyclohexenones: Synthesis and Kinase Activity Assays of Cyclopropyl-Fused Cyclohexane Nucleosides. | Jurado, S., et al. 2022. Int J Mol Sci. 23: PMID: 36077100
- Conformationally Locked Carbocyclic Nucleosides Built on a 4'-Hydroxymethyl-3'-hydroxybicyclo[4.1.0]heptane Template. Stereoselective Synthesis and Antiviral Activity. | Jurado, S., et al. 2022. J Org Chem. 87: 15166-15177. PMID: 36300902
- Facile Preparation of a Bispherical Silver-Carbon Photocatalyst and Its Enhanced Degradation Efficiency of Methylene Blue, Rhodamine B, and Methyl Orange under UV Light. | Islam, MA., et al. 2022. Nanomaterials (Basel). 12: PMID: 36432244
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Silver cyanate, 5 g | sc-272459 | 5 g | $46.00 |
