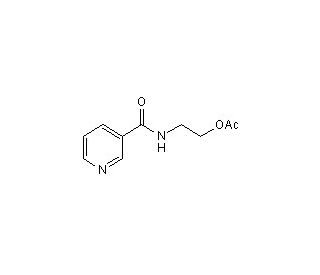
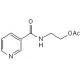
Molecular structure of SG 209, CAS Number: 83440-03-3
SG 209 (CAS 83440-03-3)
Alternate Names:
N-[2-(Acetoxy)ethyl]-3-pyridinecarboxamide
Application:
SG 209 is a K+-channel opener
CAS Number:
83440-03-3
Molecular Weight:
208.22
Molecular Formula:
C10H12N2O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
SG 209 is a chemical that functions as a potent inhibitor of a specific enzyme involved in cellular signaling pathways. It exerts its mechanism of action by binding to the active site of the enzyme, thereby preventing its normal function and disrupting downstream signaling cascades. This disruption ultimately leads to the modulation of various cellular processes, including gene expression, cell proliferation, and apoptosis. SG 209′s mechanism of action involves the interference with key protein-protein interactions, leading to the inhibition of specific cellular responses. Sg 209 plays a role in elucidating the signaling networks within cells and is instrumental in uncovering potential targets.
SG 209 (CAS 83440-03-3) References
- Purification of a neuroprotective component of Parawixia bistriata spider venom that enhances glutamate uptake. | Fontana, AC., et al. 2003. Br J Pharmacol. 139: 1297-309. PMID: 12890709
- Nicorandil inhibits the release of TNFalpha from a lymphocyte cell line and peripheral blood lymphocytes. | Wei, XM., et al. 2003. Int Immunopharmacol. 3: 1581-8. PMID: 14555283
- The effect of a nicorandil congener on isolated human myometrium. | Parimala, K., et al. 2006. Eur J Obstet Gynecol Reprod Biol. 126: 176-9. PMID: 16216408
- 2-Nicotinamidoethyl acetate (SG-209) is a potassium channel opener: structure activity relationship among nicorandil derivatives. | Ishibashi, T., et al. 1991. Naunyn Schmiedebergs Arch Pharmacol. 344: 235-9. PMID: 1658666
- The group at C2 of N-ethylnicotinamide determines the vasodilator potencies and mechanisms of action of nicorandil and its congeners in canine coronary arteries. | Satoh, K., et al. 1991. Naunyn Schmiedebergs Arch Pharmacol. 344: 589-95. PMID: 1839805
- ATP-sensitive potassium channel activation induces angiogenesis in vitro and in vivo. | Umaru, B., et al. 2015. J Pharmacol Exp Ther. 354: 79-87. PMID: 25977483
- The modulatory role of prime identified compounds in the bioactive fraction of Homalium zeylanicum in high-fat diet fed-streptozotocin-induced type 2 diabetic rats. | Rout, D., et al. 2020. J Ethnopharmacol. 260: 113099. PMID: 32535241
- A further study of the vasodilator and negative inotropic mechanisms of action of nicorandil and its congeners in the canine heart. | Satoh, K., et al. 1994. Cardiovasc Drugs Ther. 8: 227-34. PMID: 7918135
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
SG 209, 10 mg | sc-204276 | 10 mg | $94.00 | |||
SG 209, 50 mg | sc-204276A | 50 mg | $411.00 |
