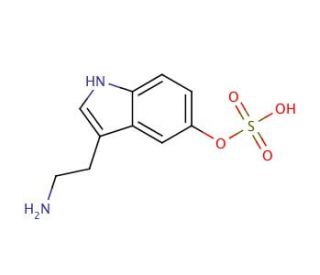
Serotonin O-Sulfate (CAS 16310-20-6)
See product citations (1)
QUICK LINKS
Serotonin O-sulfate, also known as 5-hydroxytryptamine-O-sulfate or 5-HT-O-S, holds a significant position as an essential metabolite of serotonin. Serotonin, a neurotransmitter,undergoes transformation to produce this important compound. The enzyme 5Serotonin O-sulfate (5-HT-OST) facilitates its formation, playing a role in various biochemical processes. Moreover, serotonin O-sulfate serves as a key intermediate in the metabolism of serotonin and other neurotransmitters. The precise mechanism of action of serotonin O-sulfate is not fully elucidated. However, it is believed to function as a regulator of the serotonin system by modulating the activity of serotonin receptors. Additionally, it has been suggested that serotonin O-sulfate may act as a modulator of the serotonin transporter, responsible for the uptake of serotonin.
Serotonin O-Sulfate (CAS 16310-20-6) References
- Interactions of tryptamine derivatives with serotonin transporter species variants implicate transmembrane domain I in substrate recognition. | Adkins, EM., et al. 2001. Mol Pharmacol. 59: 514-23. PMID: 11179447
- Serotonin catabolism depends upon location of release: characterization of sulfated and gamma-glutamylated serotonin metabolites in Aplysia californica. | Stuart, JN., et al. 2003. J Neurochem. 84: 1358-66. PMID: 12614336
- Synthesis and properties of serotonin O-sulfate. | KISHIMOTO, Y., et al. 1961. J Biochem. 49: 436-40. PMID: 13756366
- Recent advances in the application of capillary electrophoresis to neuroscience. | Powell, PR. and Ewing, AG. 2005. Anal Bioanal Chem. 382: 581-91. PMID: 15726336
- Contributions of capillary electrophoresis to neuroscience. | Lapainis, T. and Sweedler, JV. 2008. J Chromatogr A. 1184: 144-58. PMID: 18054026
- Substrate binding and translocation of the serotonin transporter studied by docking and molecular dynamics simulations. | Gabrielsen, M., et al. 2012. J Mol Model. 18: 1073-85. PMID: 21670993
- Quantification of serotonin O-sulphate by LC-MS method in plasma of healthy volunteers. | Lozda, R. and Purviņš, I. 2014. Front Pharmacol. 5: 62. PMID: 24782770
- Immunomodulatory effects mediated by serotonin. | Arreola, R., et al. 2015. J Immunol Res. 2015: 354957. PMID: 25961058
- Formation of serotonin O-sulfate by sulfotransferase of rabbit liver. | Hidaka, H., et al. 1969. Biochim Biophys Acta. 177: 354-7. PMID: 5780096
- Isolation of serotonin-O-sulfate from human urine. | Davis, VE., et al. 1966. Clin Chim Acta. 13: 380-2. PMID: 5943828
- Formation of serotonin O-sulfate by rat liver sulfating system. | Hidaka, H., et al. 1966. Arch Biochem Biophys. 117: 196-8. PMID: 5971740
- Conjugates of dopamine and serotonin in ventriculocisternal perfusates of the cat. | Hammond, DL., et al. 1984. J Neurochem. 42: 1752-7. PMID: 6726235
- Bacterial expression, purification, and characterization of a novel mouse sulfotransferase that catalyzes the sulfation of eicosanoids. | Liu, MC., et al. 1999. Biochem Biophys Res Commun. 254: 65-9. PMID: 9920733
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Serotonin O-Sulfate, 5 mg | sc-208385 | 5 mg | $396.00 |
