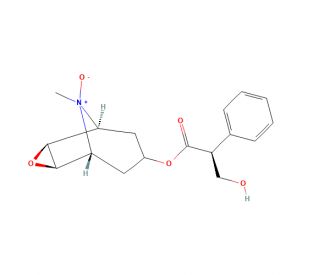
Scopolamine N-oxide (CAS 97-75-6)
QUICK LINKS
Scopolamine N-oxide is a derivative of scopolamine, also known as hyoscine, which is a naturally occurring tropane alkaloid found in plants of the Solanaceae family. Scopolamine N-oxide has been extensively studied and utilized in scientific research due to its various applications across different fields. One of the primary scientific applications of Scopolamine N-oxide is its use as a research tool in neuroscience and pharmacology. It is employed to investigate the functioning of the cholinergic system, which plays a role in cognitive processes, memory, and neurotransmission. By acting as an antagonist at muscarinic acetylcholine receptors, Scopolamine N-oxide can selectively block the activity of these receptors, enabling scientists to better understand the underlying mechanisms of various physiological and cognitive processes. The mechanism of action of Scopolamine N-oxide lies in its ability to competitively inhibit muscarinic acetylcholine receptors. By binding to these receptors, it prevents the activation of acetylcholine, a neurotransmitter involved in numerous physiological functions. This blockade leads to a reduction in cholinergic neurotransmission and subsequent effects on various organ systems, including the central nervous system.
Scopolamine N-oxide (CAS 97-75-6) References
- Modulation of the multidrug-resistance phenotype by new tropane alkaloid aromatic esters from Erythroxylum pervillei. | Silva, GL., et al. 2001. J Nat Prod. 64: 1514-20. PMID: 11754602
- Analysis of tropane and related alkaloids. | Dräger, B. 2002. J Chromatogr A. 978: 1-35. PMID: 12458942
- Three-dimensional quantitative structure-activity relationship modeling of cocaine binding by a novel human monoclonal antibody. | Paula, S., et al. 2004. J Med Chem. 47: 133-42. PMID: 14695827
- Solid-liquid extraction and cation-exchange solid-phase extraction using a mixed-mode polymeric sorbent of Datura and related alkaloids. | Mroczek, T., et al. 2006. J Chromatogr A. 1107: 9-18. PMID: 16388811
- Anticholinergic substances: A single consistent conformation. | Pauling, P. and Datta, N. 1980. Proc Natl Acad Sci U S A. 77: 708-12. PMID: 16592775
- Tropane alkaloids as medicinally useful natural products and their synthetic derivatives as new drugs. | Grynkiewicz, G. and Gadzikowska, M. 2008. Pharmacol Rep. 60: 439-63. PMID: 18799813
- Analytical approaches for the detection of epoxides and hydroperoxides in active pharmaceutical ingredients, drug products and herbals. | Elder, DP., et al. 2010. J Pharm Biomed Anal. 51: 1015-23. PMID: 20031361
- Photoactive azoimine dyes: 4-(2-pyridylazo)-N,N-diethylaniline and 4-(2-pyridylazo)-N,N-dimethylaniline: computational and experimental investigation. | Yoopensuk, S., et al. 2012. Spectrochim Acta A Mol Biomol Spectrosc. 86: 538-46. PMID: 22137011
- Bioassay-Guided Isolation of Compounds from Datura stramonium with TRAIL-Resistance Overcoming Activity. | Karmakar, UK., et al. 2016. Nat Prod Commun. 11: 185-7. PMID: 27032197
- Differentially expressed gene profile and relevant pathways of the traditional Chinese medicine cinobufotalin on MCF‑7 breast cancer cells. | Li, J., et al. 2019. Mol Med Rep. 19: 4256-4270. PMID: 30896874
- Structure of 8-methyl-8-azabicyclo[3.2.1]oct-3-yl 3,5-dichlorobenzoate methylsulfate monohydrate (MDL 72222), an antagonist at neuronal 5-HT receptors. | Carpy, A., et al. 1988. Acta Crystallogr C. 44 (Pt 3): 495-7. PMID: 3271069
- COMPARE Analysis, a Bioinformatic Approach to Accelerate Drug Repurposing against Covid-19 and Other Emerging Epidemics. | Naasani, I. 2021. SLAS Discov. 26: 345-351. PMID: 33267713
- Alkaloids of the Genus Datura: Review of a Rich Resource for Natural Product Discovery. | Cinelli, MA. and Jones, AD. 2021. Molecules. 26: PMID: 33946338
- Engineering cellular metabolite transport for biosynthesis of computationally predicted tropane alkaloid derivatives in yeast. | Srinivasan, P. and Smolke, CD. 2021. Proc Natl Acad Sci U S A. 118: PMID: 34140414
- Micellar electrokinetic chromatography of scopolamine-related anticholinergics. | Wu, HL., et al. 1998. J Chromatogr A. 802: 107-13. PMID: 9588013
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Scopolamine N-oxide, 100 mg | sc-473219 | 100 mg | $686.00 |
