Schiff′s reagent
See product citations (2)
QUICK LINKS
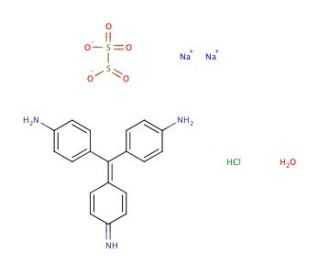
Schiff′s reagent, a classic chemical tool in histology and cytochemistry, is primarily used for the detection of aldehyde groups in biomolecules, which are often produced by the oxidation of primary alcohol groups in sugars and other compounds. The reagent is a solution of basic fuchsin decolorized by sulfurous acid, which is colorless initially but undergoes a color change upon interaction with aldehyde groups to form a magenta-colored compound. This colorimetric change is the basis for Schiff′s reagent′s utility in various staining protocols, particularly the periodic acid-Schiff (PAS) reaction, which is used to detect polysaccharides, glycoproteins, glycolipids, and other carbohydrate-containing structures in tissues. The mechanism of action involves the initial oxidation of carbohydrate moieties by periodic acid, leading to the formation of aldehyde groups. Schiff′s reagent then reacts with these aldehyde groups through an imine linkage, restoring the pink-magenta color of the fuchsin dye. This specific reaction is highly valued in research for its ability to visualize components of the cell that are not readily identifiable by other staining methods. It has been extensively utilized in studies involving glycogen storage, fungal cell wall composition, and the distribution of mucins in epithelial tissues. Moreover, Schiff′s reagent is also used in the Feulgen reaction to stain DNA by reacting with aldehyde groups generated by the acid hydrolysis of DNA, thus allowing for the visualization of nuclear material and providing insights into cell cycle dynamics and cellular morphology.
Schiff′s reagent References
- Biofilm formation by Helicobacter pylori. | Stark, RM., et al. 1999. Lett Appl Microbiol. 28: 121-6. PMID: 10063642
- Carbohydrate histochemistry studied by acetylation techniques. | McMANUS, JF. and CASON, JE. 1950. J Exp Med. 91: 651-4. PMID: 15422090
- The Use of Thionyl Chloride in the Preparation of Schiff's Reagent. | Bargee, JD. and Delamater, ED. 1948. Science. 108: 121-2. PMID: 17808736
- Examining female meiocytes of maize by confocal microscopy. | Barrell, P. and Grossniklaus, U. 2013. Methods Mol Biol. 990: 45-52. PMID: 23559201
- Hydrogen peroxide as an oxidizing agent for glycogen demonstration using Schiff's reagent in rabbit liver fixed with alcoholic and aqueous fixatives. | Abdalraheem, AB., et al. 2021. J Histotechnol. 44: 20-26. PMID: 32909929
- Visual determination of oxidation of edible oil by a nanofiber mat prepared from polyvinyl alcohol and Schiff's reagent. | Xie, M., et al. 2020. Mikrochim Acta. 187: 597. PMID: 33033874
- Qualitative Analysis of Lipid Peroxidation in Plants under Multiple Stress Through Schiff's Reagent: A Histochemical Approach. | Awasthi, JP., et al. 2018. Bio Protoc. 8: e2807. PMID: 34286024
- Gordonia sp. BSTG01 isolated from Hevea brasiliensis plantation efficiently degrades polyisoprene (rubber). | Sarkar, B. and Mandal, S. 2021. 3 Biotech. 11: 508. PMID: 34881168
- 7-Amino-4-methylcoumarin as a fluorescent substitute for Schiff's reagent: a new method that can be combined with hemalum and eosin staining on the same tissue section. | Takase, H., et al. 2023. Biotech Histochem. 98: 54-61. PMID: 35904399
- The Feulgen reaction after glutaraldehyde fixation. | Kasten, FH. and Lala, R. 1975. Stain Technol. 50: 197-201. PMID: 49954
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Schiff′s reagent, 500 ml | sc-301793 | 500 ml | $112.00 |
