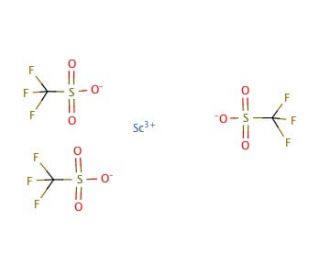
Molecular structure of Scandium(III) triflate, CAS Number: 144026-79-9
Scandium(III) triflate (CAS 144026-79-9)
Alternate Names:
Sc(OTf)3
Application:
Scandium(III) triflate is an efficient acylation catalyst
CAS Number:
144026-79-9
Molecular Weight:
492.16
Molecular Formula:
C3F9O9S3Sc
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Scandium(III) triflate is an extremely active, efficient, recoverable and reusable acylation catalyst. This is an important catalyst in Friedel-Crafts acylation, Diels-Alder reactions, and other carbon-carbon bond-forming reactions. It stereochemically catalyzes the radical. It is used in the synthesis of organic compounds, such as carboxylic acids, alcohols, and heterocyclic compounds, as well as inorganic compounds, such as metal oxides and metal halides. Scandium(III) triflate is also used in the synthesis of metal-organic frameworks (MOFs).
Scandium(III) triflate (CAS 144026-79-9) References
- Enantioselective indole Friedel--Crafts alkylations catalyzed by bis(oxazolinyl)pyridine-scandium(III) triflate complexes. | Evans, DA., et al. 2003. J Am Chem Soc. 125: 10780-1. PMID: 12952445
- Enantioselective Friedel-Crafts alkylations of alpha,beta-unsaturated 2-acyl imidazoles catalyzed by bis(oxazolinyl)pyridine-scandium(III) triflate complexes. | Evans, DA., et al. 2005. J Am Chem Soc. 127: 8942-3. PMID: 15969558
- Enantioselective aza-Diels-Alder reaction of aldimines with 'Danishefsky-type diene' catalyzed by chiral scandium(III)-N,N'-dioxide complexes. | Shang, D., et al. 2008. J Org Chem. 73: 630-7. PMID: 18088145
- Highly enantioselective one-pot, three-component Mannich-type reaction catalyzed by an N,N'-dioxide-scandium(III) complex. | Chen, S., et al. 2009. Chemistry. 15: 5884-7. PMID: 19421983
- Alkylidene malonates and α,β-unsaturated α'-hydroxyketones as practical substrates for vinylogous Friedel-Crafts alkylations in water catalysed by scandium(III) triflate/SDS. | Oelerich, J. and Roelfes, G. 2015. Org Biomol Chem. 13: 2793-9. PMID: 25604240
- Highly selective hydrothiolation of unsaturated organosilicon compounds catalyzed by scandium(III) triflate. | Kuciński, K., et al. 2015. Chemistry. 21: 4940-3. PMID: 25689836
- Scandium(iii) triflate-promoted serine/threonine-selective peptide bond cleavage. | Ni, J., et al. 2017. Chem Commun (Camb). 53: 3311-3314. PMID: 28144647
- Stereocontrolled glycoside synthesis by activation of glycosyl sulfone donors with scandium(iii) triflate. | Xolin, A., et al. 2018. Org Biomol Chem. 16: 325-335. PMID: 29260828
- Functionalized Cyclopentanes via Sc(III)-Catalyzed Intramolecular Enolate Alkylation. | McAtee, CC., et al. 2018. Tetrahedron. 74: 3306-3313. PMID: 30262933
- Predominant cleavage of proteins N-terminal to serines and threonines using scandium(III) triflate. | Koehler, CJ. and Thiede, B. 2020. J Biol Inorg Chem. 25: 61-66. PMID: 31667593
- Scandium(III) Triflate Catalyzed Direct Synthesis of N-Unprotected Ketimines. | Kondo, Y., et al. 2020. Org Lett. 22: 120-125. PMID: 31854997
- Synthesis and antimicrobial properties of new chitosan derivatives containing guanidinium groups. | Salama, A., et al. 2020. Carbohydr Polym. 241: 116363. PMID: 32507164
- Scandium(III) Triflate-Catalyzed Reaction of Aroyl-Substituted Donor-Acceptor Cyclopropanes with 1-Naphthylamines: Access to Dibenzo[c,h]acridines. | Thangamani, M. and Srinivasan, K. 2021. J Org Chem. 86: 1172-1177. PMID: 33236912
- Dielectric properties of chitosan and two ionic derivatives: Effect of counter anions. | Salama, A., et al. 2022. Carbohydr Polym. 297: 120018. PMID: 36184170
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Scandium(III) triflate, 250 mg | sc-253543 | 250 mg | $27.00 | |||
Scandium(III) triflate, 1 g | sc-253543A | 1 g | $41.00 | |||
Scandium(III) triflate, 5 g | sc-253543B | 5 g | $161.00 | |||
Scandium(III) triflate, 25 g | sc-253543C | 25 g | $437.00 |
