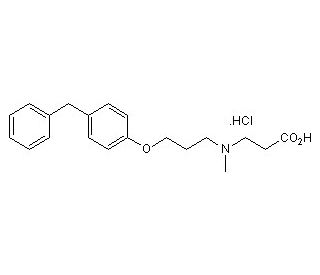
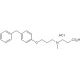
Molecular structure of SC 57461A, CAS Number: 423169-68-0
SC 57461A (CAS 423169-68-0)
See product citations (1)
Alternate Names:
3-[3-(4-benzylphenoxy)propyl-methylamino]propanoic acid hydrochloride
Application:
SC 57461A is a potent and selective inhibitor of LTA4 hydrolase
CAS Number:
423169-68-0
Purity:
≥98%
Molecular Weight:
363.88
Molecular Formula:
C20H25NO3•HCl
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
SC 57461A is a potent and selective inhibitor of LTA4 hydrolase. This compound does not inhibit other enzymes of the arachidonic acid cascade including COX-1, COX-2, LTC4 synthase and 5-lipoxygenase. SC 57461A potently inhibits LTB4 production in whole blood (IC50 = 49 nM).
SC 57461A (CAS 423169-68-0) References
- Inhibitors of leukotriene A4 (LTA4) hydrolase as potential anti-inflammatory agents. | Penning, TD. 2001. Curr Pharm Des. 7: 163-79. PMID: 11311111
- Pharmacological characterization of SC-57461A (3-[methyl[3-[4-(phenylmethyl)phenoxy]propyl]amino]propanoic acid HCl), a potent and selective inhibitor of leukotriene A(4) hydrolase I: in vitro studies. | Askonas, LJ., et al. 2002. J Pharmacol Exp Ther. 300: 577-82. PMID: 11805219
- Pharmacological characterization of SC-57461A (3-[methyl[3-[4-(phenylmethyl)phenoxy]propyl]amino]propanoic acid HCl), a potent and selective inhibitor of leukotriene A(4) hydrolase II: in vivo studies. | Kachur, JF., et al. 2002. J Pharmacol Exp Ther. 300: 583-7. PMID: 11805220
- Synthesis of potent leukotriene A(4) hydrolase inhibitors. Identification of 3-[methyl[3-[4-(phenylmethyl)phenoxy]propyl]amino]propanoic acid. | Penning, TD., et al. 2002. J Med Chem. 45: 3482-90. PMID: 12139459
- Pyrrolidine and piperidine analogues of SC-57461A as potent, orally active inhibitors of leukotriene A(4) hydrolase. | Penning, TD., et al. 2002. Bioorg Med Chem Lett. 12: 3383-6. PMID: 12419366
- Mechanisms of intimal hyperplasia learned from a murine carotid artery ligation model. | Zhang, LN., et al. 2008. Curr Vasc Pharmacol. 6: 37-43. PMID: 18220938
- Tandem Benzophenone Amino Pyridines, Potent and Selective Inhibitors of Human Leukotriene C4 Synthase. | Kleinschmidt, TK., et al. 2015. J Pharmacol Exp Ther. 355: 108-16. PMID: 26283693
- Dual anti-inflammatory and selective inhibition mechanism of leukotriene A4 hydrolase/aminopeptidase: insights from comparative molecular dynamics and binding free energy analyses. | Appiah-Kubi, P. and Soliman, ME. 2016. J Biomol Struct Dyn. 34: 2418-33. PMID: 26555301
- The leukotriene B4-leukotriene B4 receptor axis promotes cisplatin-induced acute kidney injury by modulating neutrophil recruitment. | Deng, B., et al. 2017. Kidney Int. 92: 89-100. PMID: 28318626
- Inhibition of leukotriene B4 synthesis protects against early brain injury possibly via reducing the neutrophil-generated inflammatory response and oxidative stress after subarachnoid hemorrhage in rats. | Ye, ZN., et al. 2018. Behav Brain Res. 339: 19-27. PMID: 29133197
- 5-lipoxygenase-dependent biosynthesis of novel 20:4 n-3 metabolites with anti-inflammatory activity. | Gagnon, KJ., et al. 2018. Prostaglandins Leukot Essent Fatty Acids. 138: 38-44. PMID: 30392579
- Label-free target identification reveals oxidative DNA damage as the mechanism of a selective cytotoxic agent. | Park, H. and Park, SB. 2019. Chem Sci. 10: 3449-3458. PMID: 30996934
- Isolation and identification of metabolites of leukotriene A4 hydrolase inhibitor SC-57461 in rats. | Yuan, JH., et al. 1996. Drug Metab Dispos. 24: 1124-33. PMID: 8894515
Inhibitor of:
Enzyme, LT-β, LTA4H, and LTB4R2.Activator of:
LTA4H.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
SC 57461A, 5 mg | sc-204266 | 5 mg | $151.00 | |||
SC 57461A, 25 mg | sc-204266A | 25 mg | $577.00 |
