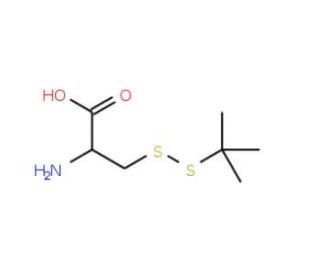
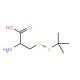
Molecular structure of S-tert-Butylmercapto-L-cysteine, CAS Number: 30044-51-0
S-tert-Butylmercapto-L-cysteine (CAS 30044-51-0)
Alternate Names:
3-(tert-Butyldithio)-L-alanine; S-(tert-Butylthio)-L-cysteine
CAS Number:
30044-51-0
Molecular Weight:
209.33
Molecular Formula:
C7H15NO2S2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
S-tert-butylmercapto-L-cysteine (S-BMC) is an important S-protected L-cysteine derivative for peptide synthesis. It is used as a synthetic reagent for other compounds, as a ligand for metal ions, and as a substrate for enzymes. It is also used to study protein structure and function. Laboratory studies have found that S-tert-Butylmercapto-L-cysteine inhibits bacterial and fungal growth and inhibits the production of reactive oxygen species. It also has anti-inflammatory and anticancer effects.
S-tert-Butylmercapto-L-cysteine (CAS 30044-51-0) References
- Influence of the Topology of Poly(L-Cysteine) on the Self-Assembly, Encapsulation and Release Profile of Doxorubicin on Dual-Responsive Hybrid Polypeptides. | Stavroulaki, D., et al. 2023. Pharmaceutics. 15: PMID: 36986652
- How controlled and versatile is N-carboxy anhydride (NCA) polymerization at 0 C? Effect of temperature on homo-, block-and graft (co) polymerization | Habraken, G. J., Peeters, M., Dietz, C. H., Koning, C. E., & Heise, A. 2010. Polymer Chemistry. 1(4): 514-524.
- Comb-like poly(l-cysteine) derivatives with different side groups: synthesis via photochemistry and click chemistry, multi-responsive nanostructures, triggered drug release and cytotoxicity | Wu, X., Zhou, L., Su, Y., & Dong, C. M. 2015. Polymer Chemistry. 6(38): 6857-6869.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
S-tert-Butylmercapto-L-cysteine, 5 g | sc-255527 | 5 g | $95.00 |
