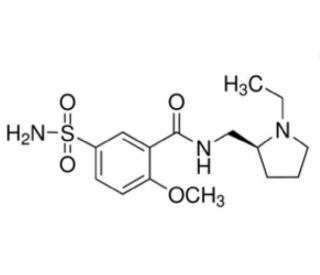
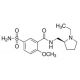
(S)-(−)-Sulpiride (CAS 23672-07-3)
See product citations (1)
QUICK LINKS
(S)-(−)-Sulpiride is a compound of interest in the field of neurochemistry and animal behavior research. It is recognized for its selective antagonistic effects on dopamine D2 receptors, which makes it a useful tool in the study of dopaminergic signaling pathways. By using (S)-(−)-Sulpiride, researchers can investigate the role of dopamine in regulating mood, movement, and hormone secretion in certain models. This compound is also utilized in biochemical assays to elucidate the mechanisms of receptor-ligand interactions within the central nervous system. Additionally, (S)-(−)-Sulpiride serves as a reference compound in the synthesis and characterization of new chemical entities targeting dopaminergic systems. Its enantiomerically pure form allows for the exploration of chiral influences on biological activity and receptor specificity.
(S)-(−)-Sulpiride (CAS 23672-07-3) References
- R(+)-8-OH-DPAT, a serotonin(1A) receptor agonist, potentiated S(-)-sulpiride-induced dopamine release in rat medial prefrontal cortex and nucleus accumbens but not striatum. | Ichikawa, J. and Meltzer, HY. 1999. J Pharmacol Exp Ther. 291: 1227-32. PMID: 10565846
- The effects of S(-) and R(+) sulpiride, metoclopramide, cisapride and domperidone on the small intestine suggest DA2-receptors are involved in the control of small intestinal transit time in rats. | Zuccato, E., et al. 1992. Pharmacol Res. 26: 179-85. PMID: 1409258
- Behavioral effects of D1 and D2 dopamine receptor antagonists in squirrel monkeys. | Bergman, J., et al. 1991. J Pharmacol Exp Ther. 258: 910-7. PMID: 1890625
- Dopamine D2High receptors stimulated by phencyclidines, lysergic acid diethylamide, salvinorin A, and modafinil. | Seeman, P., et al. 2009. Synapse. 63: 698-704. PMID: 19391150
- Rat juxtaglomerular cells are endowed with DA-1 dopamine receptors mediating renin release. | Kurtz, A., et al. 1988. J Cardiovasc Pharmacol. 12: 658-63. PMID: 2467083
- Activation of DA1 receptors by dopamine or fenoldopam increases cyclic AMP levels in the renal artery but not in the superior cervical ganglion of the rat. | Alkadhi, KA., et al. 1986. J Pharmacol Exp Ther. 238: 547-53. PMID: 2874213
- Biochemical and pharmacological characterization of ganglionic dopamine receptors. | Sabouni, MH., et al. 1987. Clin Exp Hypertens A. 9: 873-87. PMID: 2887312
- Presynaptic inhibitory dopamine receptors on noradrenergic nerve terminals: analysis of biphasic actions of dopamine and apomorphine on the release of endogenous norepinephrine in rat hypothalamic slices. | Misu, Y., et al. 1985. J Pharmacol Exp Ther. 235: 771-7. PMID: 3001276
- Preparation and characterization of a chiral molecularly imprinted polymer with a novel functional monomer for controlled release of S-sulpiride. | Wang, L., et al. 2021. Int J Pharm. 601: 120526. PMID: 33781882
- Presynaptic dopamine receptors in the cardiovascular system. | Langer, SZ., et al. 1987. Clin Exp Hypertens A. 9: 837-51. PMID: 3621629
- Effects of dopamine and its agonists and antagonists on the receptive field properties of ganglion cells in the rabbit retina. | Jensen, RJ. and Daw, NW. 1986. Neuroscience. 17: 837-55. PMID: 3703255
- Effects of dopamine, N-N-di-n-propyl dopamine, and (R)- and (S)-sulpiride on guinea pig blood pressure. | Horn, PT., et al. 1982. J Cardiovasc Pharmacol. 4: 668-75. PMID: 6181343
- Changes in sensitivity of release modulating dopamine autoreceptors after chronic treatment with haloperidol. | Nowak, JZ., et al. 1983. J Pharmacol Exp Ther. 226: 558-64. PMID: 6875865
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-(−)-Sulpiride, 5 g | sc-258111 | 5 g | $79.00 |
