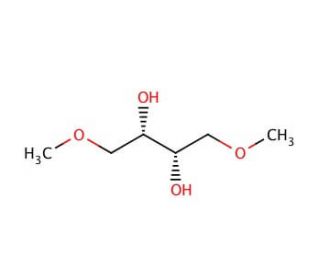
(S,S)-(−)-1,4-Dimethoxy-2,3-butanediol (CAS 50622-10-1)
QUICK LINKS
(S,S)-(−)-1,4-Dimethoxy-2,3-butanediol functions as a chiral building block in organic synthesis, particularly in the production of intermediates and chemicals. Its mechanism of action involves participating in asymmetric synthesis reactions, where its chiral nature allows for the creation of enantiomerically pure compounds. (S,S)-(−)-1,4-Dimethoxy-2,3-Butanediol serves as a component in the development of new chemical entities and materials, contributing to the advancement of synthetic methodologies and the production of complex molecules with high stereochemical control. Its role in the experimental applications involves enabling the creation of structurally diverse compounds with specific stereochemical properties. At the molecular level, (S,S)-(−)-1,4-Dimethoxy-2,3-butanediol interacts with other reagents and catalysts to facilitate the formation of chiral centers, ultimately leading to the synthesis of valuable chemical intermediates.
(S,S)-(−)-1,4-Dimethoxy-2,3-butanediol (CAS 50622-10-1) References
- Methodologies for Analyzing Soluble Organic Compounds in Extraterrestrial Samples: Amino Acids, Amines, Monocarboxylic Acids, Aldehydes, and Ketones. | Simkus, DN., et al. 2019. Life (Basel). 9: PMID: 31174308
- Analyses of Aliphatic Aldehydes and Ketones in Carbonaceous Chondrites. | Aponte, JC., et al. 2019. ACS Earth Space Chem. 3: 463-472. PMID: 32617450
- Aliphatic Aldehydes in the Earth's Crust-Remains of Prebiotic Chemistry? | Großmann, Y., et al. 2022. Life (Basel). 12: PMID: 35888015
- A stereodivergent construction of β-lactam skeletons via condensation of ester enolates and a chiral imine | Fujisawa, T., Ukaji, Y., Noro, T., Date, K., & Shimizu, M. 1991. Tetrahedron letters. 32(51): 7563-7566.
- Successive 1,4- and 1,2-additions of organometallic reagents to a chiral binaphthyl ester: one step synthesis of optically active ketones | Fuji, K., Tanaka, K., Mizuchi, M., & Hosoi, S. 1991. Tetrahedron letters. 32(49): 7277-7280.
- FT ICR investigations of chiral supramolecular propellers of dialkyltartrate trimers with methylammonium ions | Denisov, E. V., Shustryakov, V., Nikolaev, E. N., Winkler, F. J., & Medina, R. 1997. International journal of mass spectrometry and ion processes. 167: 259-268.
- Asymmetric synthesis of amines by nucleophilic 1,2-addition of organometallic reagents to the CN-double bond | Enders, D., & Reinhold, U. 1997. Tetrahedron: Asymmetry. 8(12): 1895-1946.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S,S)-(−)-1,4-Dimethoxy-2,3-butanediol, 1 g | sc-253456 | 1 g | $250.00 |
