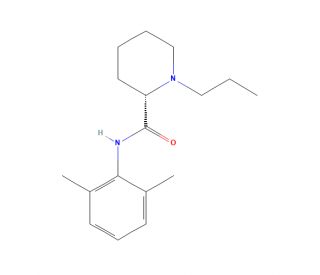
Molecular structure of (S)-Ropivacaine
(S)-Ropivacaine (CAS 84057-95-4)
Alternate Names:
(S)-N-(2,6-Dimethylphenyl)-1-propyl-2-piperidinecarboxamide
CAS Number:
84057-95-4
Molecular Weight:
274.40
Molecular Formula:
C17H26N2O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
(S)-Ropivacaine acts on the sodium channels in neuronal cells, blocking the conduction of nerve impulses. This results in a loss of sensation in the affected area, providing analgesic and anesthetic effects. (S)-Ropivacaine may act to cause decreases in neuronal excitability, calcium influx into the cell, and the release of neurotransmitters, such as glutamate and norepinephrine. It has been used in a variety of in vitro applications, including cell culture studies, enzyme assays, and receptor binding studies. These studies have found anti-inflammatory, anticonvulsant, antinociceptive, and anti-allodynic effects of (S)-Ropivacaine.
(S)-Ropivacaine (CAS 84057-95-4) References
- Determination of the enantiomeric purity of S-ropivacaine by capillary electrophoresis with methyl-beta-cyclodextrin as chiral selector using conventional and complete filling techniques. | Amini, A., et al. 1999. Eur J Pharm Sci. 9: 17-24. PMID: 10493992
- Ropivacaine attenuates pulmonary vasoconstriction induced by thromboxane A2 analogue in the isolated perfused rat lung. | Fischer, LG., et al. 2000. Reg Anesth Pain Med. 25: 187-94. PMID: 10746533
- The differential effects of stereoisomers of ropivacaine and bupivacaine on cerebral pial arterioles in dogs. | Iida, H., et al. 2001. Anesth Analg. 93: 1552-6, table of contents. PMID: 11726442
- Differences in cardiotoxicity of bupivacaine and ropivacaine are the result of physicochemical and stereoselective properties. | Graf, BM., et al. 2002. Anesthesiology. 96: 1427-34. PMID: 12170056
- Interaction of ropivacaine with cloned cardiac Kv4.3/KChIP2.2 complexes. | Friederich, P. and Solth, A. 2004. Anesthesiology. 101: 1347-56. PMID: 15564942
- Enantioselective actions of bupivacaine and ropivacaine on coronary vascular resistance at cardiotoxic concentrations. | Burmester, MD., et al. 2005. Anesth Analg. 100: 707-712. PMID: 15728056
- Differential effects of bupivacaine and ropivacaine enantiomers on intracellular Ca2+ regulation in murine skeletal muscle fibers. | Zink, W., et al. 2005. Anesthesiology. 102: 793-8. PMID: 15791109
- Direct effect of ropivacaine involves lipoxygenase pathway activation in rat aortic smooth muscle. | Sung, HJ., et al. 2009. Can J Anaesth. 56: 298-306. PMID: 19296191
- Development and validation of a LC method for the enantiomeric purity determination of S-ropivacaine in a pharmaceutical formulation using a recently commercialized cellulose-based chiral stationary phase and polar non-aqueous mobile phase. | Dossou, KS., et al. 2011. J Pharm Biomed Anal. 54: 687-93. PMID: 21111554
- Stereostructure-based differences in the interactions of cardiotoxic local anesthetics with cholesterol-containing biomimetic membranes. | Tsuchiya, H., et al. 2011. Bioorg Med Chem. 19: 3410-5. PMID: 21550810
- Lipophilicity but not stereospecificity is a major determinant of local anaesthetic-induced cytotoxicity in human T-lymphoma cells. | Werdehausen, R., et al. 2012. Eur J Anaesthesiol. 29: 35-41. PMID: 22012177
- Do local anaesthetics interact with dihydropyridine binding sites on neuronal L-type Ca2+ channels? | Hirota, K., et al. 1997. Br J Anaesth. 78: 185-8. PMID: 9068339
- [Do 'lefthanders' make better local anesthetics? The relevance of stereoisomerism in clinical practice as shown by new local anesthetics]. | Wulf, H. 1997. Anaesthesist. 46: 622-6. PMID: 9304365
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-Ropivacaine, 100 mg | sc-472719 | 100 mg | $360.00 |
