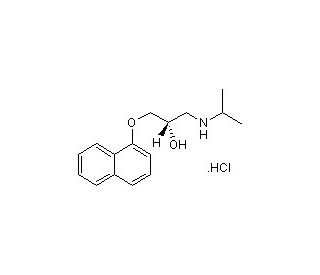
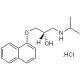
S(−)-Propranolol hydrochloride (CAS 4199-10-4)
See product citations (4)
QUICK LINKS
S(−)-Propranolol hydrochloride is a small molecule beta-AR (beta-adrenergic) receptor antagonist and the active isomer of (±)-Propranolol preparations. S(−)-Propranolol hydrochloride blocks the binding of epinephrine, norepinephrine, and other endogenous catecholamines to the beta-adrenergic receptor, impeding increases in cardiac flow velocity and general stimulation of the sympathetic nervous system signaled by association of these molecules to the beta-adrenergic receptor. In addition to blockade of agonist binding, antagonism of the beta-adrenergic receptor by S(−)-Propranolol hydrochloride produces negative chronotropic and inotropic action, effectively dampening the force and rate of cardiac contraction. These negative chronotropic and inotropic effects correlate to a demonstrated suppression of adrenaline-induced cardiac arrhythmia by S(−)-Propranolol hydrochloride. Suppression of beta-adrenergic receptor activation by S(−)-Propranolol hydrochloride has been widely exploited in counteracting situations sensitive to heightened cardiac activity including hypertension, angina pectoris, and cardiac ischemia. S(−)-Propranolol Hydrochloride is an inhibitor of SR-1A, SR-1B and SR-2C.
S(−)-Propranolol hydrochloride (CAS 4199-10-4) References
- Synthesis, stereoselective enzymatic hydrolysis, and skin permeation of diastereomeric propranolol ester prodrugs. | Udata, C., et al. 1999. J Pharm Sci. 88: 544-50. PMID: 10229647
- Stereospecific pharmacokinetics and pharmacodynamics of beta-adrenergic blockers in humans. | Mehvar, R. and Brocks, DR. 2001. J Pharm Pharm Sci. 4: 185-200. PMID: 11466176
- Solubility, metastable zone width, and racemic characterization of propranolol hydrochloride. | Wang, X., et al. 2002. Chirality. 14: 318-24. PMID: 11968072
- Inhibition of (-)-propranolol hydrochloride by its enantiomer in white mice. | Kuzeff, RM., et al. 2003. Forsch Komplementarmed Klass Naturheilkd. 10: 309-14. PMID: 14707479
- Inhibition of (-)-propranolol hydrochloride by its enantiomer in white mice--a placebo-controlled randomized study. | Kuzeff, RM., et al. 2004. Forsch Komplementarmed Klass Naturheilkd. 11: 14-9. PMID: 15004443
- Enantiospecific toxicity of the beta-blocker propranolol to Daphnia magna and Pimephales promelas. | Stanley, JK., et al. 2006. Environ Toxicol Chem. 25: 1780-6. PMID: 16833138
- Semipreparative enantiomer separation of propranolol hydrochloride by high-performance liquid chromatography using cellulose tris(3,5-Dimethylphenylcarbamate) chiral stationary phase. | Chen, L., et al. 2008. J Chromatogr Sci. 46: 767-71. PMID: 19007476
- Chiral interactions of the drug propranolol and α1-acid-glycoprotein at a micro liquid-liquid interface. | Lopes, P. and Kataky, R. 2012. Anal Chem. 84: 2299-304. PMID: 22250754
- Effects of (R)- and (S)-propranolol hydrochloride enantiomers on the resonance Rayleigh scattering spectra with erythrosine B as probe and their analytical applications. | Yang, J., et al. 2015. Talanta. 134: 754-760. PMID: 25618732
- Study on the removal of organic micropollutants from aqueous and ethanol solutions by HAP membranes with tunable hydrophilicity and hydrophobicity. | He, J., et al. 2017. Chemosphere. 174: 380-389. PMID: 28187384
- Novel chiral metal organic frameworks functionalized composites for facile preparation of optically pure propranolol hydrochlorides. | Ma, X., et al. 2019. J Pharm Biomed Anal. 172: 50-57. PMID: 31026772
- Optical isomers of propranolol. | Howe, R. and Shanks, RG. 1966. Nature. 210: 1336-8. PMID: 5963764
- Enantioseparation of beta-blockers labelled with a chiral fluorescent reagent, R (-)-DBD-PyNCS, by reversed-phase liquid chromatography. | Toyo'oka, T., et al. 1997. J Pharm Biomed Anal. 15: 1467-76. PMID: 9226577
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
S(−)-Propranolol hydrochloride, 100 mg | sc-200153 | 100 mg | $104.00 |
