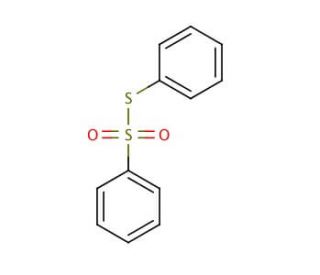
S-Phenyl benzenethiosulfonate (CAS 1212-08-4)
QUICK LINKS
S-Phenyl benzenethiosulfonate exhibits solubility in water, alcohol, and various organic solvents. Its versatile nature allows it to serve multiple purposes, including protein and peptide labeling, enzyme substrate utilization, and utilization as a reagent in organic synthesis. Moreover, it finds applications in studying the structure and functionality of proteins and other biological molecules, as well as investigating the impact of drugs and toxins on cellular systems. S-Phenyl benzenethiosulfonate is proposed to function as a substrate for enzymes and engage in interactions with proteins and other cellular constituents. These interactions are hypothesized to involve electrostatic interactions, hydrogen bonding, and hydrogen-bonded interactions with other cellular molecules.
S-Phenyl benzenethiosulfonate (CAS 1212-08-4) References
- Iodide-Catalyzed Synthesis of Secondary Thiocarbamates from Isocyanides and Thiosulfonates. | Mampuys, P., et al. 2016. Org Lett. 18: 2808-11. PMID: 27276236
- Amine Activation: Synthesis of N-(Hetero)arylamides from Isothioureas and Carboxylic Acids. | Zhu, YP., et al. 2016. Org Lett. 18: 4602-5. PMID: 27570902
- Synthesis of Secondary Amides from Thiocarbamates. | Mampuys, P., et al. 2018. Org Lett. 20: 4235-4239. PMID: 29975068
- Gas chromatography-mass spectrometric identification of cyanide using a nucleophilic substitution based derivatization with S-phenyl benzenethiosulfonate. | Goud, DR., et al. 2020. Anal Methods. 12: 5839-5845. PMID: 33227115
- Visible-light-promoted synthesis of secondary and tertiary thiocarbamates from thiosulfonates and N-substituted formamides. | Bi, WZ., et al. 2021. Org Biomol Chem. 19: 8701-8705. PMID: 34590107
- Photoexcited sulfenylation of C(sp3)-H bonds in amides using thiosulfonates. | Bi, WZ., et al. 2022. Org Biomol Chem. 20: 3902-3906. PMID: 35502883
- Phenyl trifluorovinyl sulfide: a radical acceptor for preparation of gem-difluoromethylene compounds | Okano, T., Chokai, M., Hiraishi, M., Yoshizawa, M., Kusukawa, T., & Fujita, M. 2004. Tetrahedron. 60(18): 4031-4035.
- Oxidation of organic sulfides and disulfides with a tert-butyl hydroperoxide-aluminum tri-tert-butoxide system | Zaburdaeva, E. A., & Dodonov, V. A. 2011. Russian Chemical Bulletin. 60: 185-187.
- Molecular and vibrational structure of thiosulfonate S-esters | TXT Luu, F Duus, J Spanget-Larsen. 2013. Journal of Molecular Structure. 1049: 165-171.
- Amine Activation: N‐Arylamino Acid Amide Synthesis from Isothioureas and Amino Acids | Zhu, Y. P., Mampuys, P., Sergeyev, S., Ballet, S., & Maes, B. U. 2017. Advanced Synthesis & Catalysis. 359(14): 2481-2498.
- Solvent effect on the electrochemical reduction of S-phenyl benzenethiosulfonate | Hamed, E. M. 2018. Research on Chemical Intermediates. 44: 1847-1864.
- Synthesis of Vinyl Sulfones via I2‐mediated Alkene Sulfonylations with Thiosulfonates | Hwang, S. J., Shyam, P. K., & Jang, H. Y. 2018. Bulletin of the Korean Chemical Society. 39(4): 535-539.
- External Oxidant-Free Oxidative Tandem Cyclization: NaI-Catalyzed Thiolation for the Synthesis of 3-Thiosubstituted Pyrroles | and Bingxiang Yuan, Yong Jiang, Zhenjie Qi, Xin Guan, Ting Wang, Rulong Yan. 2019. Advanced Synthesis & Catalysis. 361(22): 5112-5117.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
S-Phenyl benzenethiosulfonate, 1 g | sc-229161 | 1 g | $209.00 |
