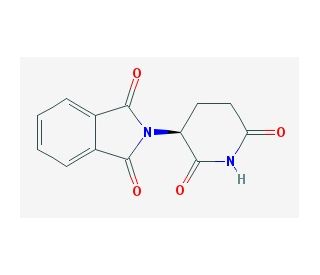
(S)-(−)-Thalidomide (CAS 841-67-8)
QUICK LINKS
(S)-(−)-Thalidomide is an optically active isomer of Thalidomide. Inhibits FGF-induced angiogenesis and replication of human immunodeficiency virus type 1. (S)-thalidomide is a 2-(2,6-dioxopiperidin-3-yl)-1H-isoindole-1,3(2H)-dione that has S-configuration at the chiral centre. It has a role as a teratogenic agent. It is an enantiomer of a (R)-thalidomide. It enhances TNF-α production induced by phorbol 12-myristate 13-acetate (TPA). Understanding how (S)-(−)-thalidomide interacts with biological targets, such as proteins or DNA, can provide valuable insights into the molecular mechanisms underlying its effects. This research can contribute to the broader knowledge of how small molecules influence cellular processes and lead to the discovery of new targets. The synthesis of pure enantiomers like (S)-(−)-thalidomide is a significant challenge in organic chemistry. Developing methods for the enantioselective synthesis or the resolution of racemic mixtures into their individual enantiomers is for producing compounds with the desired biological activity.
(S)-(−)-Thalidomide (CAS 841-67-8) References
- In vitro biotransformation of (R)- and (S)-thalidomide: application of circular dichroism spectroscopy to the stereochemical characterization of the hydroxylated metabolites. | Meyring, M., et al. 2002. Anal Chem. 74: 3726-35. PMID: 12175160
- s-thalidomide has a greater effect on apoptosis than angiogenesis in a multiple myeloma cell line. | Liu, WM., et al. 2004. Hematol J. 5: 247-54. PMID: 15167912
- Chiral discrimination between thalidomide enantiomers using a solid surface with two-dimensional chirality. | Nakanishi, T., et al. 2004. Chirality. 16 Suppl: S36-9. PMID: 15239071
- Recent advances in analytical determination of thalidomide and its metabolites. | Bosch, ME., et al. 2008. J Pharm Biomed Anal. 46: 9-17. PMID: 18023317
- A new method for determination of both thalidomide enantiomers using HPLC systems. | Sembongi, K., et al. 2008. Biol Pharm Bull. 31: 497-500. PMID: 18310917
- Thalidomide-a notorious sedative to a wonder anticancer drug. | Zhou, S., et al. 2013. Curr Med Chem. 20: 4102-8. PMID: 23931282
- Towards understanding the interaction of (S)-thalidomide with nucleobases. | Baranowska-Łączkowska, A., et al. 2020. Arch Biochem Biophys. 693: 108566. PMID: 32896516
- The structure of isolated thalidomide as reference for its chirality-dependent biological activity: a laser-ablation rotational study. | Blanco, S., et al. 2021. Phys Chem Chem Phys. 23: 13705-13713. PMID: 34128013
- A comparison of the teratogenic activity of thalidomide in rabbits and rats. | Schumacher, H., et al. 1968. J Pharmacol Exp Ther. 160: 189-200. PMID: 5639104
- Thalidomide is an inhibitor of angiogenesis. | D'Amato, RJ., et al. 1994. Proc Natl Acad Sci U S A. 91: 4082-5. PMID: 7513432
- Thalidomide inhibits the replication of human immunodeficiency virus type 1. | Makonkawkeyoon, S., et al. 1993. Proc Natl Acad Sci U S A. 90: 5974-8. PMID: 8327469
- Enantiomers of thalidomide: blood distribution and the influence of serum albumin on chiral inversion and hydrolysis. | Eriksson, T., et al. 1998. Chirality. 10: 223-8. PMID: 9499573
- Evaluation of stability difference between asymmetric homochiral dimer in (S)-thalidomide crystal and symmetric heterochiral dimer in (RS)-thalidomide crystal | Toshiya Suzuki, Masahito Tanaka, Motoo Shiro, Norio Shibata, Tetsuya Osaka & Toru Asahi. 2010. Phase Transitions. 83: 223-234.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-(−)-Thalidomide, 10 mg | sc-208337 | 10 mg | $203.00 | |||
(S)-(−)-Thalidomide, 50 mg | sc-208337A | 50 mg | $681.00 |
