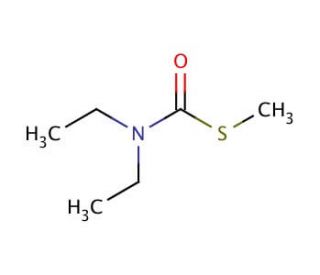
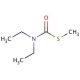
S-Methyl-N,N-diethylthiocarbamate (CAS 37174-63-3)
QUICK LINKS
S-Methyl-N,N-diethylthiocarbamate (SMDETC) is a colorless liquid compound with a distinct odor, widely utilized in diverse areas of scientific research. This compound exhibits miscibility in water and finds application in various scientific disciplines, namely biochemistry, physiology, and pharmacology. Its effectiveness in studying the impacts of chemicals, environmental toxins, hormones, and other molecules on the human body has been extensively documented through laboratory experiments. Although the precise mechanism of action of S-Methyl-N,N-diethylthiocarbamate remains partially elusive, it is believed to operate by inhibiting specific enzymes, including acetylcholinesterase, responsible for acetylcholine breakdown. Furthermore, S-Methyl-N,N-diethylthiocarbamate is thought to engage with fatty acids and hormones, leading to a diverse array of biochemical and physiological effects.
S-Methyl-N,N-diethylthiocarbamate (CAS 37174-63-3) References
- Disulfiram metabolite S-methyl-N,N-diethylthiocarbamate quantitation in human plasma with reverse phase ultra performance liquid chromatography and mass spectrometry. | Hochreiter, J., et al. 2012. J Chromatogr B Analyt Technol Biomed Life Sci. 897: 80-4. PMID: 22534656
- Antitubercular activity of disulfiram, an antialcoholism drug, against multidrug- and extensively drug-resistant Mycobacterium tuberculosis isolates. | Horita, Y., et al. 2012. Antimicrob Agents Chemother. 56: 4140-5. PMID: 22615274
- Reactivity and binding mode of disulfiram, its metabolites, and derivatives in SARS-CoV-2 PLpro: insights from computational chemistry studies. | Nogara, PA., et al. 2022. J Mol Model. 28: 354. PMID: 36222962
- Peptide alpha-amidation: differential regulation by disulfiram and its metabolite, diethyldithiocarbamate. | Mueller, GP. and Altarac, M. 1995. Neuropeptides. 28: 333-40. PMID: 7666952
- Aldehyde dehydrogenase of mice inhibited by thiocarbamate herbicides. | Quistad, GB., et al. 1994. Life Sci. 55: 1537-44. PMID: 7968224
- Inhibition of aldehyde dehydrogenase by disulfiram and its metabolite methyl diethylthiocarbamoyl-sulfoxide. | Veverka, KA., et al. 1997. Biochem Pharmacol. 53: 511-8. PMID: 9105402
- Inhibition of human mitochondrial aldehyde dehydrogenase by the disulfiram metabolite S-methyl-N,N-diethylthiocarbamoyl sulfoxide: structural characterization of the enzyme adduct by HPLC-tandem mass spectrometry. | Tomlinson, AJ., et al. 1997. Biochem Pharmacol. 54: 1253-60. PMID: 9416976
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
S-Methyl-N,N-diethylthiocarbamate, 10 mg | sc-212820 | 10 mg | $449.00 | |||
S-Methyl-N,N-diethylthiocarbamate, 50 mg | sc-212820A | 50 mg | $1224.00 | |||
S-Methyl-N,N-diethylthiocarbamate, 100 mg | sc-212820B | 100 mg | $2244.00 |
