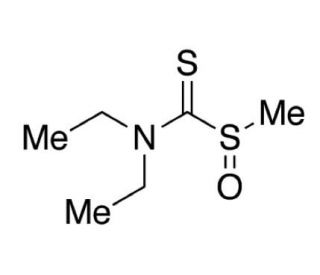
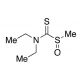
S-Methyl-N,N-diethyldithiocarbamate Sulfoxide (CAS 145195-14-8)
See product citations (1)
QUICK LINKS
S-Methyl-N,N-diethyldithiocarbamate Sulfoxide, also known as N,N-diethyl-1-methylsulfinylmethanethioamide, is a human mitochondrial aldehyde dehydrogenase inhibitor as well as a metabolite of Disulfiram(sc-205654A). S-Methyl-N,N-diethyldithiocarbamate Sulfoxide, a synthetic compound classified as a thiocarbamate herbicide, is widely employed in agriculture to manage weed proliferation in various crops like rice, wheat, and maize. Beyond its herbicidal role, this compound has garnered significant attention in scientific research. Extensive studies have revealed its potential in inhibiting the growth of cancer cells in controlled laboratory applications. Moreover, it has been instrumental in inducing oxidative stress in animal models to comprehend cellular damage and repair mechanisms. Furthermore, S-Methyl-N,N-diethyldithiocarbamate Sulfoxide has proven valuable in the development of biosensors utilized for detecting heavy metal contaminants in environmental samples. Its diverse applications span both agricultural practices and scientific investigations, contributing to advancements in crop management and environmental monitoring.
S-Methyl-N,N-diethyldithiocarbamate Sulfoxide (CAS 145195-14-8) References
- Aldehyde dehydrogenase inhibitors: a comprehensive review of the pharmacology, mechanism of action, substrate specificity, and clinical application. | Koppaka, V., et al. 2012. Pharmacol Rev. 64: 520-39. PMID: 22544865
- N-acetyl-S-(N,N-diethylcarbamoyl) cysteine in rat nucleus accumbens, medial prefrontal cortex, and in rat and human plasma after disulfiram administration. | Winefield, RD., et al. 2015. J Pharm Biomed Anal. 107: 518-25. PMID: 25720821
- Mechanisms of Neuronal Protection against Excitotoxicity, Endoplasmic Reticulum Stress, and Mitochondrial Dysfunction in Stroke and Neurodegenerative Diseases. | Prentice, H., et al. 2015. Oxid Med Cell Longev. 2015: 964518. PMID: 26576229
- Involvement of endoplasmic reticulum stress in formalin-induced pain is attenuated by 4-phenylbutyric acid. | Zhou, F., et al. 2017. J Pain Res. 10: 653-662. PMID: 28360534
- Novel therapeutic features of disulfiram against hepatocellular carcinoma cells with inhibitory effects on a disintegrin and metalloproteinase 10. | Goto, K., et al. 2018. Oncotarget. 9: 18821-18831. PMID: 29721164
- Disulfiram-loaded mixed nanoparticles with high drug-loading and plasma stability by reducing the core crystallinity for intravenous delivery. | Zhuo, X., et al. 2018. J Colloid Interface Sci. 529: 34-43. PMID: 29883928
- Population Pharmacokinetics and Pharmacodynamics of Disulfiram on Inducing Latent HIV-1 Transcription in a Phase IIb Trial. | Lee, SA., et al. 2019. Clin Pharmacol Ther. 105: 692-702. PMID: 30137649
- Binding Affinity of Some Endogenous and Synthetic TSPO Ligands Regarding the rs6971 Polymorphism. | Berroterán-Infante, N., et al. 2019. Int J Mol Sci. 20: PMID: 30699908
- Endoplasmic Reticulum Stress and the Unfolded Protein Response in Cerebral Ischemia/Reperfusion Injury. | Wang, L., et al. 2022. Front Cell Neurosci. 16: 864426. PMID: 35602556
- UPR Responsive Genes Manf and Xbp1 in Stroke. | Lõhelaid, H., et al. 2022. Front Cell Neurosci. 16: 900725. PMID: 35783104
- Isothiocyanates (ITCs) 1-(Isothiocyanatomethyl)-4-phenylbenzene and 1-Isothiocyanato-3,5-bis(trifluoromethyl)benzene-Aldehyde Dehydrogenase (ALDH) Inhibitors, Decreases Cisplatin Tolerance and Migratory Ability of NSCLC. | Kryczka, J., et al. 2022. Int J Mol Sci. 23: PMID: 35955773
- Reactivity and binding mode of disulfiram, its metabolites, and derivatives in SARS-CoV-2 PLpro: insights from computational chemistry studies. | Nogara, PA., et al. 2022. J Mol Model. 28: 354. PMID: 36222962
- Studies on the metabolic activation of disulfiram in rat. Evidence for electrophilic S-oxygenated metabolites as inhibitors of aldehyde dehydrogenase and precursors of urinary N-acetylcysteine conjugates. | Hu, P., et al. 1997. J Pharmacol Exp Ther. 281: 611-7. PMID: 9152363
- Inhibition of recombinant human mitochondrial aldehyde dehydrogenase by two intermediate metabolites of disulfiram. | Mays, DC., et al. 1998. Biochem Pharmacol. 55: 1099-103. PMID: 9605433
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
S-Methyl-N,N-diethyldithiocarbamate Sulfoxide, 10 mg | sc-483485 | 10 mg | $965.00 |
