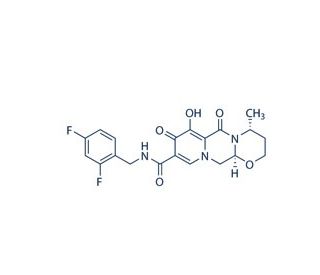
S/GSK1349572 (CAS 1051375-16-6)
QUICK LINKS
S/GSK1349572 inhibits recombinant HIV-1 integrase-catalyzed strand transfer in vitro. S/GSK1349572 potently inhibits HIV replication in cells infected with a self-inactivating PHIV lentiviral vector, such as peripheral blood mononuclear cells (PBMCs), MT-4 cells and CIP4. S/GSK1349572 exhibits potent activity against five different nonnucleoside reverse transcription inhibitor--resistant or nucleoside reverse transcription inhibitor--resistant viruses in vitro. S/GSK1349572 shows equivalent activity against two protease inhibitor-resistant viruses similarly to that against wild-type virus. S/GSK1349572 is an integrase inhibitor, which means it works by blocking the integration of HIV-1 DNA into the host cell genome. This prevents the virus from replicating and spreading.
S/GSK1349572 (CAS 1051375-16-6) References
- S/GSK1349572, a new integrase inhibitor for the treatment of HIV: promises and challenges. | Lenz, JC. and Rockstroh, JK. 2011. Expert Opin Investig Drugs. 20: 537-48. PMID: 21381981
- Resistance associated mutations to dolutegravir (S/GSK1349572) in HIV-infected patients--impact of HIV subtypes and prior raltegravir experience. | Garrido, C., et al. 2011. Antiviral Res. 90: 164-7. PMID: 21439330
- Prevalence of resistance mutations related to integrase inhibitor S/GSK1349572 in HIV-1 subtype B raltegravir-naive and -treated patients. | Malet, I., et al. 2011. J Antimicrob Chemother. 66: 1481-3. PMID: 21474479
- Structural and functional analyses of the second-generation integrase strand transfer inhibitor dolutegravir (S/GSK1349572). | Hare, S., et al. 2011. Mol Pharmacol. 80: 565-72. PMID: 21719464
- Dolutegravir (S/GSK1349572) exhibits significantly slower dissociation than raltegravir and elvitegravir from wild-type and integrase inhibitor-resistant HIV-1 integrase-DNA complexes. | Hightower, KE., et al. 2011. Antimicrob Agents Chemother. 55: 4552-9. PMID: 21807982
- Cross-resistance profile of the novel integrase inhibitor Dolutegravir (S/GSK1349572) using clonal viral variants selected in patients failing raltegravir. | Canducci, F., et al. 2011. J Infect Dis. 204: 1811-5. PMID: 21984737
- Prevalent polymorphisms in wild-type HIV-1 integrase are unlikely to engender drug resistance to dolutegravir (S/GSK1349572). | Vavro, C., et al. 2013. Antimicrob Agents Chemother. 57: 1379-84. PMID: 23295935
- Carbamoyl pyridone HIV-1 integrase inhibitors 3. A diastereomeric approach to chiral nonracemic tricyclic ring systems and the discovery of dolutegravir (S/GSK1349572) and (S/GSK1265744). | Johns, BA., et al. 2013. J Med Chem. 56: 5901-16. PMID: 23845180
- Dolutegravir: first global approval. | Ballantyne, AD. and Perry, CM. 2013. Drugs. 73: 1627-37. PMID: 24052331
- Dolutegravir, a second-generation integrase inhibitor for the treatment of HIV-1 infection. | Rathbun, RC., et al. 2014. Ann Pharmacother. 48: 395-403. PMID: 24259658
- Might dolutegravir be part of a functional cure for HIV? | Wainberg, MA., et al. 2016. Can J Microbiol. 62: 375-82. PMID: 27031127
- Dolutegravir/Lamivudine Single-Tablet Regimen: A Review in HIV-1 Infection. | Scott, LJ. 2020. Drugs. 80: 61-72. PMID: 31865558
- Pharmacokinetics and placental transfer of dolutegravir in pregnancy. | Ikumi, NM., et al. 2022. J Antimicrob Chemother. 77: 283-289. PMID: 34618029
- An up-to-date evaluation of dolutegravir/abacavir/lamivudine for the treatment of HIV. | Christensen, BL. and Tan, DH. 2022. Expert Opin Pharmacother. 23: 439-446. PMID: 35073817
- Dolutegravir in Pregnancy as Compared with Current HIV Regimens in the United States. | Patel, K., et al. 2022. N Engl J Med. 387: 799-809. PMID: 36053505
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
S/GSK1349572, 5 mg | sc-364605 | 5 mg | $374.00 | |||
S/GSK1349572, 50 mg | sc-364605B | 50 mg | $1514.00 | |||
S/GSK1349572, 200 mg | sc-364605A | 200 mg | $4214.00 |
