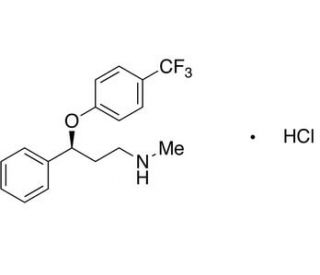
(S)-Fluoxetine Hydrochloride (CAS 114247-06-2)
QUICK LINKS
(S)-Fluoxetine Hydrochloride is a compound utilized in various areas of research, focusing on its interaction with neurotransmitter systems. It is primarily used in studies of the serotonin transporter, where it serves as a selective inhibitor, allowing scientists to investigate the kinetics and dynamics of serotonin reuptake at synaptic junctions. In neurochemical research, (S)-Fluoxetine Hydrochloride is employed to understand the modulation of serotonin levels in the brain. This compound is also useful in examining the structural and functional aspects of monoamine transporters and their role in neurotransmitter regulation. Additionally, it is applied in models to observe the effects of serotonin reuptake inhibition on various physiological and behavioral parameters.
(S)-Fluoxetine Hydrochloride (CAS 114247-06-2) References
- Preparation of polymer-supported Ru-TsDPEN catalysts and use for enantioselective synthesis of (S)-fluoxetine. | Li, Y., et al. 2005. Org Biomol Chem. 3: 2513-8. PMID: 15999182
- Enantiospecific sublethal effects of the antidepressant fluoxetine to a model aquatic vertebrate and invertebrate. | Stanley, JK., et al. 2007. Chemosphere. 69: 9-16. PMID: 17582462
- A validated enantioselective assay for the simultaneous quantitation of (R)-, (S)-fluoxetine and (R)-, (S)-norfluoxetine in ovine plasma using liquid chromatography with tandem mass spectrometry (LC/MS/MS). | Chow, TW., et al. 2011. J Chromatogr B Analyt Technol Biomed Life Sci. 879: 349-58. PMID: 21242112
- Trace analysis of fluoxetine and its metabolite norfluoxetine. Part II: Enantioselective quantification and studies of matrix effects in raw and treated wastewater by solid phase extraction and liquid chromatography-tandem mass spectrometry. | Barclay, VK., et al. 2012. J Chromatogr A. 1227: 105-14. PMID: 22265784
- Simultaneous enantioselective quantification of fluoxetine and norfluoxetine in human milk by direct sample injection using 2-dimensional liquid chromatography-tandem mass spectrometry. | Alvim, J., et al. 2016. J Chromatogr A. 1451: 120-126. PMID: 27208983
- Absolute configurations and pharmacological activities of the optical isomers of fluoxetine, a selective serotonin-uptake inhibitor. | Robertson, DW., et al. 1988. J Med Chem. 31: 1412-7. PMID: 3260286
- Clinical serum metabolomics study on fluoxetine hydrochloride for depression. | Shen, D., et al. 2021. Neurosci Lett. 746: 135585. PMID: 33352278
- Prozac (fluoxetine, Lilly 110140), the first selective serotonin uptake inhibitor and an antidepressant drug: twenty years since its first publication. | Wong, DT., et al. 1995. Life Sci. 57: 411-41. PMID: 7623609
- Fluoxetine selectively alters 5-hydroxytryptamine1A and gamma-aminobutyric acidB receptor-mediated hyperpolarization in area CA1, but not area CA3, hippocampal pyramidal cells. | Beck, SG., et al. 1997. J Pharmacol Exp Ther. 281: 115-22. PMID: 9103487
- Neurotransmitter receptor and transporter binding profile of antidepressants and their metabolites. | Owens, MJ., et al. 1997. J Pharmacol Exp Ther. 283: 1305-22. PMID: 9400006
- Determination of fluoxetine hydrochloride enantiomeric excess using high-performance liquid chromatography with chiral stationary phases. | Olsen, BA., et al. 1998. J Pharm Biomed Anal. 17: 623-30. PMID: 9682145
- Enantioselective synthesis of (S)- and (R)-fluoxetine hydrochloride | William H Miles, Elizabeth J Fialcowitz, E Scott Halstead. 2001. Tetrahedron. 57: 9925-9929.
- Pd-Catalyzed kinetic resolution of benzylic alcohols: a practical synthesis of (R)-tomoxetine and (S)-fluoxetine hydrochlorides | Iliyas Sayyed Ali, Arumugam Sudalai. 2002. Tetrahedron Letters. 43: 5435-5436.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-Fluoxetine Hydrochloride, 5 mg | sc-208367 | 5 mg | $307.00 |
