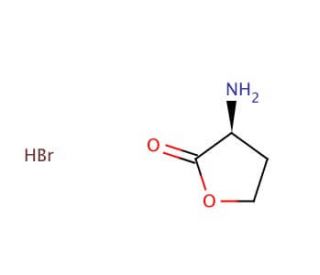
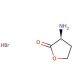
(S)-(−)-α-Amino-γ-butyrolactone hydrobromide (CAS 15295-77-9)
QUICK LINKS
(S)-(−)-α-Amino-γ-butyrolactone hydrobromide is a vital organic compound extensively employed in diverse scientific research applications. Its significance lies in serving as a synthetic intermediate for the preparation of a wide range of compounds. It serves as a fundamental starting material for synthesizing an array of compounds, including chiral amines, aminoalcohols, and amino acids. Additionally, it acts as a reagent in organic synthesis and functions as a ligand in asymmetric catalysis. The mechanism of action underlying (S)-(−)-α-Amino-γ-butyrolactone hydrobromide is not yet fully elucidated. However, it is believed to involve the formation of a transient intermediate, which subsequently undergoes conversion into the desired product. This intermediate arises from the reaction between bromine, base, and the dihydrofuran-2-one.
(S)-(−)-α-Amino-γ-butyrolactone hydrobromide (CAS 15295-77-9) References
- Degradation of N-acylhomoserine lactones, the bacterial quorum-sensing molecules, by acylase. | Xu, F., et al. 2003. J Biotechnol. 101: 89-96. PMID: 12523973
- The Pseudomonas aeruginosa autoinducer N-3-oxododecanoyl homoserine lactone accelerates apoptosis in macrophages and neutrophils. | Tateda, K., et al. 2003. Infect Immun. 71: 5785-93. PMID: 14500500
- The quorum-quenching lactonase from Bacillus thuringiensis is a metalloprotein. | Thomas, PW., et al. 2005. Biochemistry. 44: 7559-69. PMID: 15895999
- Structure and specificity of a quorum-quenching lactonase (AiiB) from Agrobacterium tumefaciens. | Liu, D., et al. 2007. Biochemistry. 46: 11789-99. PMID: 17900178
- Absolute configuration and antimicrobial activity of acylhomoserine lactones. | Pomini, AM. and Marsaioli, AJ. 2008. J Nat Prod. 71: 1032-6. PMID: 18465897
- Mechanism of the quorum-quenching lactonase (AiiA) from Bacillus thuringiensis. 2. Substrate modeling and active site mutations. | Momb, J., et al. 2008. Biochemistry. 47: 7715-25. PMID: 18627130
- Microwave synthesis and evaluation of phenacylhomoserine lactones as anticancer compounds that minimally activate quorum sensing pathways in Pseudomonas aeruginosa. | Oliver, CM., et al. 2009. J Med Chem. 52: 1569-75. PMID: 19260689
- Long-chain acyl-homoserine lactones from Methylobacterium mesophilicum: synthesis and absolute configuration. | Pomini, AM., et al. 2009. J Nat Prod. 72: 2125-9. PMID: 19919062
- Discovery of a quorum sensing modulator pharmacophore by 3D small-molecule microarray screening. | Marsden, DM., et al. 2010. Org Biomol Chem. 8: 5313-23. PMID: 20886127
- Total synthesis of the hypermodified RNA bases wybutosine and hydroxywybutosine and their quantification together with other modified RNA bases in plant materials. | Hienzsch, A., et al. 2013. Chemistry. 19: 4244-8. PMID: 23417961
- Metabolic pathway of inorganic and organic selenocompounds labeled with stable isotope in Japanese quail. | Anan, Y., et al. 2014. Anal Bioanal Chem. 406: 7959-66. PMID: 25326891
- Synthesis and antiviral evaluation of a novel series of homoserine-based inhibitors of the hepatitis C virus NS3/4A serine protease. | Alexandre, FR., et al. 2015. Bioorg Med Chem Lett. 25: 3984-91. PMID: 26231161
- Direct detection of small molecules using a nano-molecular imprinted polymer receptor and a quartz crystal resonator driven at a fixed frequency and amplitude. | Guha, A., et al. 2020. Biosens Bioelectron. 158: 112176. PMID: 32275209
- Generic sensor platform based on electro-responsive molecularly imprinted polymer nanoparticles (e-NanoMIPs). | Garcia-Cruz, A., et al. 2020. Microsyst Nanoeng. 6: 83. PMID: 34567693
- Pseudopeptide inhibitors of Ras farnesyl-protein transferase. | Graham, SL., et al. 1994. J Med Chem. 37: 725-32. PMID: 8145221
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-(−)-α-Amino-γ-butyrolactone hydrobromide, 1 g | sc-253483 | 1 g | $42.00 |
