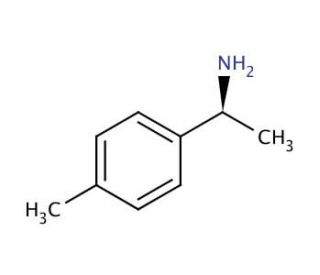
Molecular structure of (S)-(−)-α,4-Dimethylbenzylamine, CAS Number: 27298-98-2
(S)-(−)-α,4-Dimethylbenzylamine (CAS 27298-98-2)
CAS Number:
27298-98-2
Molecular Weight:
135.21
Molecular Formula:
C9H13N
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
(S)-(−)-α,4-Dimethylbenzylamine has been used in the study of heterogeneous Raney nickel and cobalt catalysts for racemization and dynamic kinetic resolution of amines. This compound has displayed photochemical reactions under uv irradiation and high-pressure conditions, which are the basis for its use for diffraction studies. Using x-ray diffraction studies or x-ray crystallography to monitor and analyze the crystals, gaining valuable insights into their structure and molecular parameters.
(S)-(−)-α,4-Dimethylbenzylamine (CAS 27298-98-2) References
- Reversed-phase high-performance liquid chromatographic enantioresolution of six beta-blockers using dinitrophenyl-L-Pro-N-hydroxysuccinimide ester, N-succinimidyl-(S)-2-(6-methoxynaphth-2-yl) propionate and twelve variants of Sanger's reagent as chiral derivatizing reagents. | Bhushan, R. and Tanwar, S. 2009. Biomed Chromatogr. 23: 1291-9. PMID: 19488982
- Synthesis of dinitrophenyl-L-Pro-N-hydroxysuccinimide ester and four new variants of Sanger's reagent having chiral amines and their application for enantioresolution of mexiletine using reversed-phase high-performance liquid chromatography. | Bhushan, R. and Tanwar, S. 2009. J Chromatogr A. 1216: 5769-73. PMID: 19552908
- Enantiomeric purity of chiral derivatizing reagents for enantioresolution. | Bhushan, R. 2011. Bioanalysis. 3: 2057-60. PMID: 21942516
- HPLC enantioresolution of (R,S)-baclofen using three newly synthesized dichloro-s-triazine reagents having amines and five others having amino acids as chiral auxiliaries. | Bhushan, R. and Dixit, S. 2012. Biomed Chromatogr. 26: 743-8. PMID: 21989982
- Synthesis of new chiral xanthone derivatives acting as nerve conduction blockers in the rat sciatic nerve. | Fernandes, C., et al. 2012. Eur J Med Chem. 55: 1-11. PMID: 22819594
- Chiral derivatizations applied for the separation of unusual amino acid enantiomers by liquid chromatography and related techniques. | Ilisz, I., et al. 2013. J Chromatogr A. 1296: 119-39. PMID: 23598164
- A novel approach for enantioseparation as applied to (RS)-etodolac from pharmaceutical formulations: LC MS and density functional theory support for confirmation of diastereomers so separated. | Singh, M. and Bhushan, R. 2015. Biomed Chromatogr. 29: 1330-7. PMID: 25641127
- Enantioresolution of (RS)-baclofen by liquid chromatography: A review. | Batra, S. and Bhushan, R. 2017. Biomed Chromatogr. 31: PMID: 27591736
- Bioassay, determination and separation of enantiomers of atenolol by direct and indirect approaches using liquid chromatography: A review. | Batra, S. and Bhushan, R. 2018. Biomed Chromatogr. 32: PMID: 28905405
- Enantioseparation of (RS)-fexofenadine and enhanced detection as the diastereomeric amide and anhydride derivatives using liquid chromatography-mass spectrometry. | Malik, P. and Bhushan, R. 2018. Biomed Chromatogr. 32: e4217. PMID: 29601646
- Methods and approaches for determination and enantioseparation of (RS)-propranolol. | Batra, S. and Bhushan, R. 2019. Biomed Chromatogr. 33: e4370. PMID: 30121955
- Liquid chromatographic methods for separation, determination, and bioassay of enantiomers of etodolac: A review. | Singh, M., et al. 2020. J Sep Sci. 43: 18-30. PMID: 31389172
- Assessment and application of Marfey's reagent and analogs in enantioseparation: a decade's perspective. | Sethi, S., et al. 2021. Biomed Chromatogr. 35: e4990. PMID: 32959901
- Progress in the Enantioseparation of β-Blockers by Chromatographic Methods. | Yang, Y., et al. 2021. Molecules. 26: PMID: 33477385
- 'Ab Ovo' Chiral Phases and Chiral Reagents for Liquid Chromatographic Separation and Isolation of Enantiomers. | Bhushan, R. 2022. Chem Rec. 22: e202100295. PMID: 35005840
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-(−)-α,4-Dimethylbenzylamine, 1 g | sc-236738 | 1 g | $71.00 |
