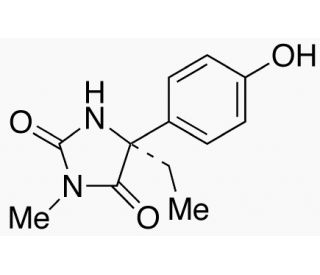
(S)-4-Hydroxy mephenytoin (CAS 82695-93-0)
See product citations (1)
QUICK LINKS
(S)-4-Hydroxy mephenytoin, a metabolite derived from the stereoselective metabolism of mephenytoin, plays a critical role in pharmacogenetic studies, particularly in the exploration of cytochrome P450 enzymes′ polymorphic nature. Its formation is predominantly catalyzed by CYP2C19, an enzyme exhibiting significant genetic variability across populations. This variability affects the metabolic clearance of substrates like (S)-4-Hydroxy mephenytoin, making it an invaluable probe in understanding the genetic factors that influence drug metabolism and enzyme activity. In research applications, the compound is utilized to assess the metabolic efficiency of CYP2C19, helping to classify individuals into different metabolizer phenotypes based on their enzyme activity levels. This classification has implications for predicting metabolic responses to a wide range of substances, thus (S)-4-Hydroxy mephenytoin serves as a key molecule in pharmacokinetic and pharmacogenomic studies, offering insights into the genetic basis of individual variations in drug metabolism.
(S)-4-Hydroxy mephenytoin (CAS 82695-93-0) References
- A model based assessment of the CYP2B6 and CYP2C19 inductive properties by artemisinin antimalarials: implications for combination regimens. | Elsherbiny, DA., et al. 2008. J Pharmacokinet Pharmacodyn. 35: 203-17. PMID: 18350255
- Chimeric mice with humanized liver: Application in drug metabolism and pharmacokinetics studies for drug discovery. | Naritomi, Y., et al. 2018. Drug Metab Pharmacokinet. 33: 31-39. PMID: 29183653
- Contributions of UDP-Glucuronosyltransferases to Human Hepatic and Intestinal Metabolism of Ticagrelor and Inhibition of UGTs and Cytochrome P450 Enzymes by Ticagrelor and its Glucuronidated Metabolite. | Liu, S., et al. 2021. Front Pharmacol. 12: 761814. PMID: 34721047
- Inhibition of cytochrome P450 enzymes and uridine 5'-diphospho-glucuronosyltransferases by vicagrel in human liver microsomes: A prediction of potential drug-drug interactions. | Liu, S., et al. 2022. Chem Biol Interact. 352: 109775. PMID: 34910929
- Capillary electrophoresis in clinical and forensic analysis. | von Heeren, F. and Thormann, W. 1997. Electrophoresis. 18: 2415-26. PMID: 9456056
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-4-Hydroxy mephenytoin, 1 mg | sc-471498A | 1 mg | $203.00 | |||
(S)-4-Hydroxy mephenytoin, 2 mg | sc-471498B | 2 mg | $275.00 | |||
(S)-4-Hydroxy mephenytoin, 5 mg | sc-471498 | 5 mg | $359.00 | |||
(S)-4-Hydroxy mephenytoin, 10 mg | sc-471498C | 10 mg | $645.00 | |||
(S)-4-Hydroxy mephenytoin, 25 mg | sc-471498D | 25 mg | $1503.00 | |||
(S)-4-Hydroxy mephenytoin, 50 mg | sc-471498E | 50 mg | $2502.00 |
