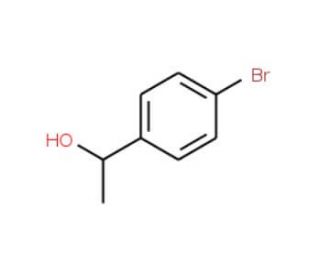
(S)-4-Bromo-α-methylbenzyl alcohol (CAS 100760-04-1)
QUICK LINKS
(S)-4-Bromo-α-methylbenzyl alcohol is an organic compound extensively used in synthesizing various compounds and chemicals. This chiral compound exists in two forms, with the (S) form being the most employed one. Its wide-ranging applications span across materials science and biochemistry. In materials science, it acts as a catalyst in organic reactions and serves as a fundamental building block for creating polymers. Its significance extends even to the field of biochemistry, where it has been extensively researched due to its intriguing biochemical effects. One fascinating aspect of (S)-4-Bromo-α-methylbenzyl alcohol lies in its ability to act as an agonist of the GABA-A receptor. Moreover, it acts as an inhibitor of acetylcholinesterase, an enzyme involved in the breakdown of the neurotransmitter acetylcholine. Additionally, 4-BP has been found to inhibit cyclooxygenase-2, another enzyme linked to the production of pro-inflammatory molecules. (S)-4-Bromo-α-methylbenzyl alcohol′s versatility and potential applications make it a highly valuable compound in diverse scientific fields, driving extensive research to uncover its promising properties and contributions to the advancement of various industries.
(S)-4-Bromo-α-methylbenzyl alcohol (CAS 100760-04-1) References
- Mechanism of asymmetric hydrogenation of ketones catalyzed by BINAP/1,2-diamine-rutheniumII complexes. | Sandoval, CA., et al. 2003. J Am Chem Soc. 125: 13490-503. PMID: 14583046
- Racemization of secondary alcohols catalyzed by cyclopentadienylruthenium complexes: evidence for an alkoxide pathway by fast beta-hydride elimination-readdition. | Martín-Matute, B., et al. 2007. Chemistry. 13: 6063-72. PMID: 17516611
- Quantitative structure enantioselective retention relationship for high-performance liquid chromatography chiral separation of 1-phenylethanol derivatives. | Szaleniec, M., et al. 2009. J Chromatogr A. 1216: 6224-35. PMID: 19631329
- Asymmetric reduction of substituted acetophenones using once immobilized Rhodotorula glutinis cells. | Kurbanoglu, EB., et al. 2010. Bioresour Technol. 101: 3825-9. PMID: 20110163
- Hot water-promoted S(N)1 solvolysis reactions of allylic and benzylic alcohols. | Xu, ZB. and Qu, J. 2013. Chemistry. 19: 314-23. PMID: 23165820
- Photochemical and Photophysical Properties of Phthalocyanines Modified with Optically Active Alcohols. | Ramos, AA., et al. 2015. Molecules. 20: 13575-90. PMID: 26213911
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-4-Bromo-α-methylbenzyl alcohol, 250 mg | sc-229257 | 250 mg | $35.00 |
