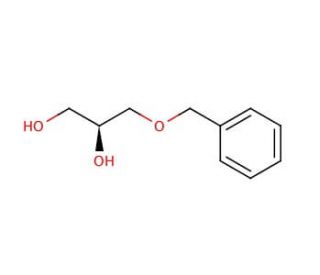
(S)-(-)-3-Benzyloxy-1,2-propanediol (CAS 17325-85-8)
QUICK LINKS
(S)-(−)-3-Benzyloxy-1,2-propanediol is a specialized chiral compound that plays a crucial role in organic synthesis, particularly in the construction of optically active molecules. This chemical features a benzyloxy group attached to a propanediol backbone, which not only provides steric hindrance but also influences the electronic environment of the molecule. Such structural attributes make it a valuable intermediate in the synthesis of complex pharmaceuticals and fine chemicals, where control of stereochemistry is paramount. In research, this compound is frequently used to study asymmetric synthesis methods, serving as a building block for developing new synthetic routes that enhance the enantioselectivity and yield of desired products. Its application in creating chiral centers is essential for producing molecules that can interact in a specific manner with biological macromolecules, advancing our understanding of molecular interactions at the chiral level. Moreover, (S)-(−)-3-Benzyloxy-1,2-propanediol′s properties are exploited in materials science, where its ability to influence the physical properties of polymers and resins is studied. The research utilizing this compound underscores its significance in synthetic chemistry, highlighting its role in innovating synthetic strategies.
(S)-(-)-3-Benzyloxy-1,2-propanediol (CAS 17325-85-8) References
- The migrastatin family: discovery of potent cell migration inhibitors by chemical synthesis. | Gaul, C., et al. 2004. J Am Chem Soc. 126: 11326-37. PMID: 15355116
- Evaluating Ylehd, a recombinant epoxide hydrolase from Yarrowia lipolytica as a potential biocatalyst for the resolution of benzyl glycidyl ether. | Bendigiri, C., et al. 2018. RSC Adv. 8: 12918-12926. PMID: 35541265
- Synthesis and triplex forming properties of an acyclic N7-glycosylated guanine nucleoside. | St Clair, A., et al. 1998. Nucleosides Nucleotides. 17: 925-37. PMID: 9708332
- Oligonucleotides containing flexible nucleoside analogs[J]. | Schneider K C, Benner S A. 1990,. Journal of the American Chemical Society,. 112(1):: 453-455.
- Preparation of O-Protected Glyceraldehydes as Building Blocks in Organic Synthesis[J]. | Dibello E, Gamenara D, Seoane G. 2015. Organic Preparations and Procedures International,., 47(6):: 415-442.
- Canela-Garayoa R. Raw and waste plant materials as sources of fungi with epoxide hydrolase activity. Application to the kinetic resolution of aryl and alkyl glycidyl ethers[J]. | Dolcet M, Torres M,. 2018,. Biocatalysis and Biotransformation,. 36(1):: 78-88.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(S)-(-)-3-Benzyloxy-1,2-propanediol, 100 mg | sc-253478 | 100 mg | $61.00 |
