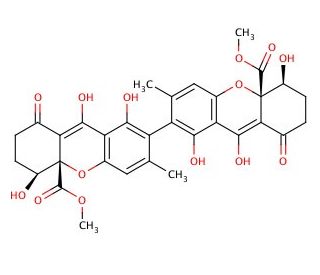
Rugulotrosin A (CAS 685135-81-3)
QUICK LINKS
Rugulotrosin A is a complex polyketide-derived natural product, distinguished by its unique chemical structure characterized by a bicyclic [2.2.2]diazaoctane core. This chemical has garnered attention in the field of natural product research due to its origin from endophytic fungi, which are known for their ability to produce bioactive secondary metabolites with novel mechanisms of action. Rugulotrosin A exhibits a mode of action primarily through the inhibition of quorum sensing, a critical communication process used by bacteria to regulate gene expression in response to population density. By disrupting this process, rugulotrosin A interferes with bacterial community behaviors, such as biofilm formation and virulence factor production, which are essential for bacterial survival and pathogenicity. This specific interaction with bacterial signaling pathways makes rugulotrosin A an interesting subject for studies focused on new strategies to mitigate bacterial infections, especially in scenarios where traditional antibiotics are ineffective due to resistance. Research involving rugulotrosin A has also extended to its potential role in modulating fungal-bacterial interactions within their natural ecosystems, providing deeper insights into the ecological roles of fungal secondary metabolites and their impact on microbial diversity and competition.
Rugulotrosin A (CAS 685135-81-3) References
- Total syntheses of secalonic acids A and D. | Qin, T. and Porco, JA. 2014. Angew Chem Int Ed Engl. 53: 3107-10. PMID: 24519991
- Atropselective syntheses of (-) and (+) rugulotrosin A utilizing point-to-axial chirality transfer. | Qin, T., et al. 2015. Nat Chem. 7: 234-40. PMID: 25698333
- Asymmetric Synthesis of Gonytolide A: Strategic Use of an Aryl Halide Blocking Group for Oxidative Coupling. | Wu, X., et al. 2018. J Am Chem Soc. 140: 5969-5975. PMID: 29658717
- The fungal gene cluster for biosynthesis of the antibacterial agent viriditoxin. | Urquhart, AS., et al. 2019. Fungal Biol Biotechnol. 6: 2. PMID: 31304040
- Fungal Diversity in Intertidal Mudflats and Abandoned Solar Salterns as a Source for Biological Resources. | Heo, YM., et al. 2019. Mar Drugs. 17: PMID: 31652878
- Asymmetric Synthesis of Rugulotrosin A. | Chen, J., et al. 2020. Org Lett. 22: 1485-1489. PMID: 32037839
- Enantioselective total synthesis of parnafungin A1 and 10a-epi-hirtusneanine. | Sun, J., et al. 2021. Chem Sci. 12: 10313-10320. PMID: 34377417
- An Enantioselective Suzuki-Miyaura Coupling To Form Axially Chiral Biphenols. | Pearce-Higgins, R., et al. 2022. J Am Chem Soc. 144: 15026-15032. PMID: 35969692
- Properties of Configurationally Stable Atropoenantiomers in Macrocyclic Natural Products and the Chrysophaentin Family. | Bewley, CA., et al. 2023. Acc Chem Res. 56: 414-424. PMID: 36731116
- Paralemnalia thyrsoides-associated fungi: phylogenetic diversity, cytotoxic potential, metabolomic profiling and docking analysis. | Taher Mohie El-Dien, R., et al. 2023. BMC Microbiol. 23: 308. PMID: 37884900
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Rugulotrosin A, 1 mg | sc-391699 | 1 mg | $213.00 |
