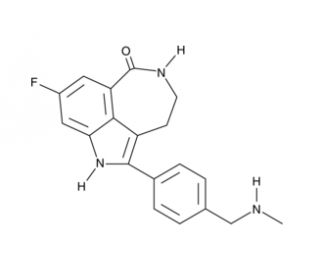
Molecular structure of Rucaparib, CAS Number: 283173-50-2
Rucaparib (CAS 283173-50-2)
Alternate Names:
8-fluoro-1,3,4,5-tetrahydro-2-[4-[(methylamino)methyl]phenyl]-6H-pyrrolo[4,3,2-ef][2]benzazepin-6-one
CAS Number:
283173-50-2
Molecular Weight:
323.4
Molecular Formula:
C19H18FN3O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Rucaparib (CAS 283173-50-2) References
- PARP Inhibitors in Metastatic Prostate Cancer: A Comprehensive Systematic Review and Meta-analysis of Existing Evidence. | Ditonno, F., et al. 2024. Clin Genitourin Cancer. 22: 402-412.e17. PMID: 38281877
- Identification of differential biological activity and synergy between the PARP inhibitor rucaparib and its major metabolite. | Hu, H., et al. 2024. Cell Chem Biol. 31: 973-988.e4. PMID: 38335967
- A Phase II Study of Rucaparib Monotherapy in Nonmetastatic, Hormone-Sensitive Prostate Cancer Demonstrating 'BRCAness' Genotype (ROAR). | Sahu, KK., et al. 2024. Oncologist. 29: 450-e725. PMID: 38452035
- ClonoScreen3D - A Novel 3-Dimensional Clonogenic Screening Platform for Identification of Radiosensitizers for Glioblastoma. | Jackson, MR., et al. 2024. Int J Radiat Oncol Biol Phys.. PMID: 38493899
- IB-DNQ and Rucaparib dual treatment alters cell cycle regulation and DNA repair in triple negative breast cancer cells. | Runnebohm, AM., et al. 2024. bioRxiv.. PMID: 38798459
- Regulatory histories of recently withdrawn ovarian cancer treatment indications of 3 PARP inhibitors in the US and Europe: lessons for the accelerated approval pathway. | Shahzad, M., et al. 2024. J Pharm Policy Pract. 17: 2351003. PMID: 38841118
- The efficacy and safety of PARP inhibitors in mCRPC with HRR mutation in second-line treatment: a systematic review and bayesian network meta-analysis. | Zhu, Q., et al. 2024. BMC Cancer. 24: 706. PMID: 38851712
- New target therapies in prostate cancer: from radioligand therapy, to PARP-inhibitors and immunotherapy. | Ceci, F., et al. 2024. Q J Nucl Med Mol Imaging. 68: 101-115. PMID: 38860274
- Financial toxicity of withdrawn poly (ADP-ribose) polymerase inhibitor indications for ovarian cancer. | Dottino, JA., et al. 2024. Value Health.. PMID: 38871026
- TRIUMPH: phase II trial of rucaparib monotherapy in patients with metastatic hormone-sensitive prostate cancer harboring germline homologous recombination repair gene mutations. | Markowski, MC., et al. 2024. Oncologist.. PMID: 38885246
- ATR, CHK1 and WEE1 inhibitors cause homologous recombination repair deficiency to induce synthetic lethality with PARP inhibitors. | Smith, HL., et al. 2024. Br J Cancer.. PMID: 38965423
- PARP inhibitor-related acute renal failure: a real-world study based on the FDA adverse event reporting system database. | Ren, X., et al. 2024. Expert Opin Drug Saf. 1-9. PMID: 38967020
- PARP inhibition with rucaparib alone followed by combination with atezolizumab: Phase Ib COUPLET clinical study in advanced gynaecological and triple-negative breast cancers. | Kristeleit, R., et al. 2024. Br J Cancer.. PMID: 38971950
Inhibitor of:
4930547N16Rik, AK7, ART2B, BABAM1, BAF53B, BC066135, BCL2L12, BRCA2, BRCAA1, C12orf32, C20orf196, C9orf102, C9orf119, CCDC98, DNAH14, DNB5, DNLZ, FAAP100, FAN, GBDR1, GTBP, H2-10, hCAP-D3, HELIC2, hMLH3, K-cadherin, KIAA1267, LPLUNC4, MEI1, MND1, Npl1, Olr1156, Olr1217, Olr170, Olr232, Olr530, Olr561, Olr659, Olr749, Ovarian Carcinoma-associated Antigen, PAMR1, PAR4, Paraxis, PARN, PARP L, PARP S, PARP-1, PARP-10, PARP-11, PARP-12, PARP-14, PARP-15, PARP-16, PARP-2, PARP-7, PARP-8, PARP-9, PIERCE 1, Pma, PMS1, PNK, PRK2, PRP19, RAD54L2, Rad57, RAP80, RECA-1, RecQL4, RFC1, RNF146, ROPN1, RuvC, TDE1, TDP2, TFIP11, TNRC18, TTYH1, V1RH17, VAP-1, XPV, XRCC2, and ZRANB3.Activator of:
4930547N16Rik, ASF1A, BTBD12, DNA pol θ, DNAH5, F730047E07Rik, FIGNL1, HPF1, KIN17, MIA3, Morc2b, MPHOSPH8, Msh6, Nibrin, pADPr, PALB2, PARG, PARP-4, PGBD5, XRCC3, and ZSWIM7.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Rucaparib, 5 mg | sc-507419 | 5 mg | $150.00 |
