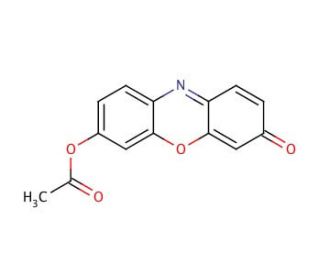
Molecular structure of Resorufin acetate, CAS Number: 1152-14-3
Resorufin acetate (CAS 1152-14-3)
Alternate Names:
7-Acetoxy-3H-phenoxazin-3-one
Application:
Resorufin acetate is a fluorogenic substrate for hydrolytic enzymes
CAS Number:
1152-14-3
Purity:
≥98%
Molecular Weight:
255.23
Molecular Formula:
C14H9NO4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Resorufin acetate is an excellent option for research purposes, owing to its stability and non-toxicity as a water-soluble compound. It is a type of fluorescent dye that becomes activated by enzymes. Enzymes catalyze the conversion of the dye to a fluorescent product upon exposure to resorufin acetate. Fluorescence spectroscopy can be utilized to identify and quantify the product.
Resorufin acetate (CAS 1152-14-3) References
- Covalent modification of sheep liver cytosolic aldehyde dehydrogenase by the oxidative addition of coloured phenoxazine, phenothiazine and phenazine derivatives. | Kitson, TM., et al. 1999. Adv Exp Med Biol. 463: 89-96. PMID: 10352673
- Mutations in distant residues moderately increase the enantioselectivity of Pseudomonas fluorescens esterase towards methyl 3bromo-2-methylpropanoate and ethyl 3phenylbutyrate. | Horsman, GP., et al. 2003. Chemistry. 9: 1933-9. PMID: 12740839
- Focusing mutations into the P. fluorescens esterase binding site increases enantioselectivity more effectively than distant mutations. | Park, S., et al. 2005. Chem Biol. 12: 45-54. PMID: 15664514
- Biochemical characterization of Alr1529, a novel SGNH hydrolase variant from Anabaena sp. PCC 7120. | Bakshy, K., et al. 2009. Biochim Biophys Acta. 1794: 324-34. PMID: 19028609
- Selective perborate signaling by deprotection of fluorescein and resorufin acetates. | Choi, MG., et al. 2010. Org Lett. 12: 1468-71. PMID: 20222739
- Synthesis and utility of fluorogenic acetoxymethyl ethers. | Lavis, LD., et al. 2011. Chem Sci. 2: 521-530. PMID: 21394227
- A conserved interdomain communication pathway of pseudosymmetrically distributed residues affects substrate specificity of the fungal multidrug transporter Cdr1p. | Kolaczkowski, M., et al. 2013. Biochim Biophys Acta. 1828: 479-90. PMID: 23122779
- The chemistry of small-molecule fluorogenic probes. | Grimm, JB., et al. 2013. Prog Mol Biol Transl Sci. 113: 1-34. PMID: 23244787
- Structural determinants of phenoxazine type compounds required to modulate the accumulation of vinblastine and vincristine in multidrug-resistant cell lines. | Thimmaiah, KN., et al. 1990. Cancer Commun. 2: 249-59. PMID: 2378785
- Antibacterial activity of resazurin-based compounds against Neisseria gonorrhoeae in vitro and in vivo. | Schmitt, DM., et al. 2016. Int J Antimicrob Agents. 48: 367-72. PMID: 27451856
- Molecular Mechanism for Isoform-Selective Inhibition of Acyl Protein Thioesterases 1 and 2 (APT1 and APT2). | Won, SJ., et al. 2016. ACS Chem Biol. 11: 3374-3382. PMID: 27748579
- Glycoengineering of Esterase Activity through Metabolic Flux-Based Modulation of Sialic Acid. | Mathew, MP., et al. 2017. Chembiochem. 18: 1204-1215. PMID: 28218815
- The action of cytosolic aldehyde dehydrogenase on resorufin acetate. | Kitson, TM. and Kitson, KE. 1997. Adv Exp Med Biol. 414: 201-8. PMID: 9059622
- Studies on the chymotrypsin-catalysed hydrolysis of resorufin acetate and resorufin bromoacetate. | Kitson, TM. 1998. Biochim Biophys Acta. 1385: 43-52. PMID: 9630505
Substrate of:
ALDH1A1, Chymotrypsin, and Enzyme.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Resorufin acetate, 25 mg | sc-208300 | 25 mg | $198.00 |
