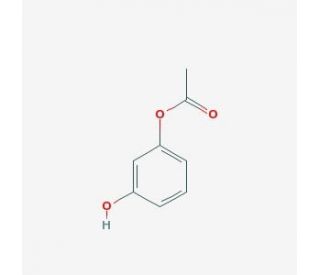
Resorcinol monoacetate (CAS 102-29-4)
QUICK LINKS
Resorcinol monoacetate is a compound that functions as a chemical intermediate in various and development processes. It acts by undergoing acetylation reactions, where the hydroxyl group of resorcinol is acetylated to form the monoacetate. Resorcinol monoacetate modification allows for the incorporation of resorcinol monoacetate into the synthesis of various organic compounds, including dyes, and polymers. At the molecular level, resorcinol monoacetate participates in acylation reactions, serving as a building block for the creation of more complex chemical structures. Its mode of action involves the formation of covalent bonds with other molecules, enabling the production of diverse chemical products. Resorcinol monoacetate plays a functional role as a versatile chemical intermediate, contributing to the synthesis of a wide range of organic compounds through its acetylation reactions.
Resorcinol monoacetate (CAS 102-29-4) References
- Discoloration of the nails; concomitant use of nail lacquer with resorcinol or resorcinol monoacetate (euresol) as cause. | LOVEMAN, AB. and FLIEGELMAN, MT. 1955. AMA Arch Derm. 72: 153-6. PMID: 14397848
- Structure-based drug design and optimization of mannoside bacterial FimH antagonists. | Han, Z., et al. 2010. J Med Chem. 53: 4779-92. PMID: 20507142
- First quantitative high-throughput screen in zebrafish identifies novel pathways for increasing pancreatic β-cell mass. | Wang, G., et al. 2015. Elife. 4: PMID: 26218223
- Novel C-Ring-Hydroxy-Substituted Controlled Deactivation Cannabinergic Analogues. | Kulkarni, S., et al. 2016. J Med Chem. 59: 6903-19. PMID: 27367336
- The Follicular Skin Microbiome in Patients With Hidradenitis Suppurativa and Healthy Controls. | Ring, HC., et al. 2017. JAMA Dermatol. 153: 897-905. PMID: 28538949
- Biocatalytic Friedel-Crafts Acylation and Fries Reaction. | Schmidt, NG., et al. 2017. Angew Chem Int Ed Engl. 56: 7615-7619. PMID: 28544673
- Collagen based biomaterials: an ideal way of increasing their resistance to infection. | Gunasekaran, S. and Chvapil, M. 1988. Biomater Artif Cells Artif Organs. 16: 771-83. PMID: 3219416
- Recent Advances in (Bio)Chemical Sensors for Food Safety and Quality Based on Silver Nanomaterials. | Ivanišević, I., et al. 2021. Food Technol Biotechnol. 59: 216-237. PMID: 34316283
- Insights from a Box-Behnken Optimization Study of Microemulsions with Salicylic Acid for Acne Therapy. | Anicescu, MC., et al. 2022. Pharmaceutics. 14: PMID: 35057071
- Identification of the metabolic remodeling profile in the early-stage of myocardial ischemia and the contributory role of mitochondrion. | He, J., et al. 2022. Bioengineered. 13: 11106-11121. PMID: 35470774
- Microwave-Assisted Extraction of Bioactive Compounds from Lentil Wastes: Antioxidant Activity Evaluation and Metabolomic Characterization. | Cavalluzzi, MM., et al. 2022. Molecules. 27: PMID: 36364300
- Treatment of Diabetes Nephropathy in Mice by Germinating Seeds of Euryale ferox through Improving Oxidative Stress. | Wang, Y., et al. 2023. Foods. 12: PMID: 36832842
- Optimization of the Brewing Conditions of Shanlan Rice Wine and Sterilization by Thermal and Intense Pulse Light. | Wu, X., et al. 2023. Molecules. 28: PMID: 37049943
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Resorcinol monoacetate, 100 g | sc-229136 | 100 g | $52.00 | |||
Resorcinol monoacetate, 500 g | sc-229136A | 500 g | $255.00 |
