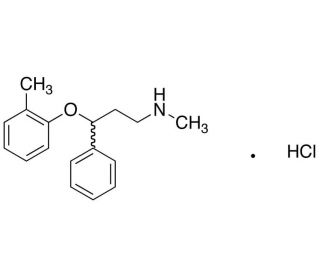
Molecular structure of rac Atomoxetine Hydrochloride, CAS Number: 82857-40-7
rac Atomoxetine Hydrochloride (CAS 82857-40-7)
Application:
rac Atomoxetine Hydrochloride is a compound active at a novel site on receptor-operated Ca2+ channels
CAS Number:
82857-40-7
Molecular Weight:
291.82
Molecular Formula:
C17H22ClNO
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
rac Atomoxetine Hydrochloride is a derivative of Tomoxetine. Tomoxetine (Atomoxetine, LY 139603; sc-204349) is a competitve and specific inhibitor of norepinephrine reuptake while demonstrating weak serotonin and dopamine inhibition as studied in synaptosomes of rat hypothalamus. Additional studies indicate that this (-) optical isomer is more effective than the (+) isomer, LY139602 (ent S-(+)-Atomoxetine Hydrochloride; sc-211408). Additionally, in the prefrontal cortex of rats, Tomoxetine was reported to not cause an increase in dopamine within the striatum or nucleus accumbens which may be useful in additional research studies investigating the reward pathways of the brain.
rac Atomoxetine Hydrochloride (CAS 82857-40-7) References
- Disposition and metabolic fate of atomoxetine hydrochloride: pharmacokinetics, metabolism, and excretion in the Fischer 344 rat and beagle dog. | Mattiuz, EL., et al. 2003. Drug Metab Dispos. 31: 88-97. PMID: 12485957
- Disposition and metabolic fate of atomoxetine hydrochloride: the role of CYP2D6 in human disposition and metabolism. | Sauer, JM., et al. 2003. Drug Metab Dispos. 31: 98-107. PMID: 12485958
- Atomoxetine hydrochloride: clinical drug-drug interaction prediction and outcome. | Sauer, JM., et al. 2004. J Pharmacol Exp Ther. 308: 410-8. PMID: 14610241
- Atomoxetine hydrochloride for the treatment of attention-deficit/hyperactivity disorder. | Caballero, J. and Nahata, MC. 2003. Clin Ther. 25: 3065-83. PMID: 14749146
- Atomoxetine hydrochloride and executive function in children with attention-deficit/hyperactivity disorder. | Barton, J., et al. 2005. J Child Adolesc Psychopharmacol. 15: 147-9. PMID: 15910196
- Children and adolescent exposures to atomoxetine hydrochloride reported to a poison control center. | Stojanovski, SD., et al. 2006. Clin Toxicol (Phila). 44: 243-7. PMID: 16749540
- Spectrophotometric Quantification of Atomoxetine Hydrochloride Based on Nucleophilic Substitution Reaction with 1,2-Naphthoquinone-4-Sulfonic Acid Sodium Salt (NQS). | Naraparaju, S., et al. 2024. Turk J Pharm Sci. 20: 405-411. PMID: 38258290
- Assessment of centanafadine in adults with attention-deficit/hyperactivity disorder: A matching-adjusted indirect comparison vs lisdexamfetamine dimesylate, atomoxetine hydrochloride, and viloxazine extended-release. | Schein, J., et al. 2024. J Manag Care Spec Pharm. 30: 528-540. PMID: 38824626
- Prozac (fluoxetine, Lilly 110140), the first selective serotonin uptake inhibitor and an antidepressant drug: twenty years since its first publication. | Wong, DT., et al. 1995. Life Sci. 57: 411-41. PMID: 7623609
- Fluoxetine selectively alters 5-hydroxytryptamine1A and gamma-aminobutyric acidB receptor-mediated hyperpolarization in area CA1, but not area CA3, hippocampal pyramidal cells. | Beck, SG., et al. 1997. J Pharmacol Exp Ther. 281: 115-22. PMID: 9103487
- Neurotransmitter receptor and transporter binding profile of antidepressants and their metabolites. | Owens, MJ., et al. 1997. J Pharmacol Exp Ther. 283: 1305-22. PMID: 9400006
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
rac Atomoxetine Hydrochloride, 5 mg | sc-212721 | 5 mg | $360.00 |
