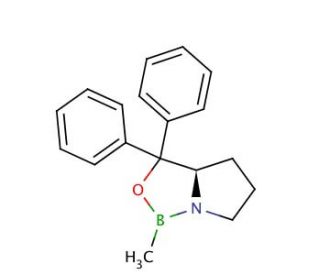
Molecular structure of (R)-(+)-2-Methyl-CBS-oxazaborolidine, CAS Number: 112022-83-0
(R)-(+)-2-Methyl-CBS-oxazaborolidine (CAS 112022-83-0)
Alternate Names:
(R)-2-Methyl-CBS-oxazaborolidine; (R)-5,5-Diphenyl-2-methyl-3,4-propano-1,3,2-oxazaborolidine; Corey's catalyst
Application:
(R)-(+)-2-Methyl-CBS-oxazaborolidine is a chemical employed in the asymmetric reduction of prochiral ketones
CAS Number:
112022-83-0
Purity:
≥96%
Molecular Weight:
277.17
Molecular Formula:
C18H20BNO
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
R)-(+)-2-Methyl-CBS-oxazaborolidine is an oxazaborolidine, which catalyzes the asymmetry in the borane reduction of prochiral ketones. Studies indicate that (R)-(+)-2-Methyl-CBS-oxazaborolidine is useful in the production of stereospecific motifs such as α-hydroxy acids, α-amino acids, symmetrical ferrocenyl diols and propargyl alcohols. We also offer (R)-(+)-oxazaborolidine (sc-258069), which can be used to synthesize enantiospecific products.
(R)-(+)-2-Methyl-CBS-oxazaborolidine (CAS 112022-83-0) References
- Asymmetric Reduction. A Convenient Method for the Reduction of Alkynyl Ketones. | Parker, KA. and Ledeboer, MW. 1996. J Org Chem. 61: 3214-3217. PMID: 11667191
- Asymmetric synthesis of corsifuran A by an enantioselective oxazaborolidine reduction. | Adams, H., et al. 2008. Org Lett. 10: 1457-60. PMID: 18336034
- Part 2. Notch-sparing γ-secretase inhibitors: The study of novel γ-amino naphthyl alcohols. | Wei, HX., et al. 2016. Bioorg Med Chem Lett. 26: 2133-7. PMID: 27020305
- A practical synthesis of xylo- and arabinofuranoside precursors by diastereoselective reduction using Corey-Bakshi-Shibata catalyst. | Utley, LM., et al. 2018. Nucleosides Nucleotides Nucleic Acids. 37: 20-34. PMID: 29336673
- Asymmetric synthesis of some chiral aryl and hetero aryl-substituted β-, γ-, δ-hydroxy esters | Hasdemir, B. 2015. Synthetic Communications. 45(9): 1082-1088.
- Stereoselective Reducing Complexes Supported in Solid Phase: Preparation and Reactivity Study | Vargas‐Durazo, J., Aguilar‐Martínez, M., Ochoa‐Terán, A., Santacruz‐Ortega, H., Alonzo, F. R., Meza, N. G., & Galvez‐Ruiz, J. C. 2019. ChemistrySelect. 4(30): 8776-8780.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(R)-(+)-2-Methyl-CBS-oxazaborolidine, 1 g | sc-258069 | 1 g | $62.00 |
