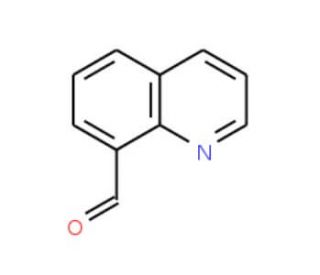
Quinoline-8-carbaldehyde (CAS 38707-70-9)
QUICK LINKS
Quinoline-8-carbaldehyde is a chemical compound that functions as a key intermediate in the synthesis of various organic compounds. Its mechanism of action involves participating in a range of chemical reactions, including condensation, oxidation, and reduction processes. Within the experimental context, Quinoline-8-carbaldehyde serves as a versatile building block for the creation of diverse molecular structures, enabling the formation of complex organic molecules. Its functional role lies in its ability to undergo specific chemical transformations, leading to the production of novel compounds with potential applications in various research fields. At the molecular level, quinoline-8-carbaldehyde interacts with other reagents to facilitate the formation of new chemical bonds, thereby contributing to the development of innovative molecules for experimental purposes. Its mechanism of action involves participating in synthetic pathways that lead to the generation of diverse chemical entities, expanding the scope of organic chemistry research.
Quinoline-8-carbaldehyde (CAS 38707-70-9) References
- Oxidation of 6- and 8-methylquinolines upon UV-illumination in the presence of a powder of TiO2 photocatalyst. | Navío, JA., et al. 2002. Photochem Photobiol Sci. 1: 133-5. PMID: 12659129
- Catalytic C(sp(3))-H Arylation of Free Primary Amines with an exo Directing Group Generated In Situ. | Xu, Y., et al. 2016. Angew Chem Int Ed Engl. 55: 9084-7. PMID: 27276342
- A pentacoordinated norbornenyl-acyl-rhodium(iii) complex as a likely intermediate in the catalytic hydroacylation of norbornadiene. | Almenara, N., et al. 2016. Dalton Trans. 45: 18502-18509. PMID: 27774563
- Modified nucleoside triphosphates exist in mammals. | Jiang, HP., et al. 2018. Chem Sci. 9: 4160-4167. PMID: 29780546
- Copper(ii) complexes based on quinoline-derived Schiff-base ligands: synthesis, characterization, HSA/DNA binding ability, and anticancer activity. | Hu, K., et al. 2018. Medchemcomm. 9: 1663-1672. PMID: 30429971
- Enhancing the kinetics of hydrazone exchange processes: an experimental and computational study. | Higgs, PL., et al. 2019. Org Biomol Chem. 17: 3218-3224. PMID: 30840013
- Cyclic acyl complexes of palladium(II). Synthesis and NMR spectroscopy of acyl complexes derived from quinoline-8-carbaldehyde and 2-(dimethylamino)benzaldehyde | C.G. Anklin, P.S. Pregosin. 1983. Journal of Organometallic Chemistry. 243: 101-109.
- Transformations of schiff bases derived from quinoline-8-carbaldehyde. Synthesis of C-8 substituted quinolines | Nüket Ocal, ÇIgdem Yolaçan, ŞEniz Kaban, Vargas M. Leonor, Vladimir Kouznetsov. 2001. Journal of Heterocyclic Chemistry. 38: 233-236.
- Functionalisation versus mineralisation of some N-heterocyclic compounds upon UV-illumination in the presence of un-doped and iron-doped TiO2 photocatalysts | J.A. Navío a, M. Macias a, M. Garcia-Gómez b, M.A. Pradera b. 2008. Applied Catalysis B: Environmental. 82: 225-232.
- Synthesis and application of new iminopyridine ligands in the enantioselective palladium-catalyzed allylic alkylation | Maurizio Solinas a, Barbara Sechi a, Giorgio Chelucci b, Salvatore Baldino b, José R. Pedro c, Gonzalo Blay c. 2014. Journal of Molecular Catalysis A: Chemical. 385: 73-77.
- Reactions of quinoline-2(6,8)-carbaldehydes with arenes by the action of various Brønsted or Lewis acids: synthesis of diarylmethylquinolines | Marina А. Borisova, Dmitry S. Ryabukhin & Aleksander V. Vasilyev. 2020. Chemistry of Heterocyclic Compounds. 56: 964–967.
- Rhodium(III)-Catalyzed Cross-Coupling of Sulfoxonium Ylides with Quinoline-8-carboxaldehydes for Synthesis of Quinoline-1,3-diketones | Xue-Li Lyu, Shi-Sheng Huang, Dr. Yuan-Qiong Huang, Prof. Dr. Hong-Jian Song, Prof. Dr. Yu-Xiu Liu, Prof. Dr. Yong-Qiang Li, Prof. Dr. Shao-Xiang Yang, Prof. Dr. Qing-Min Wang. 2021. Asian Journal of Organic Chemistry. 10: 176-179.
- Experimental and DFT studies on Hexacoordinated acyl(alkyl)and Pentacooordinated Hydroxyalkyl(phosphinite)erhodium(III). Catalytic Hydrolysis of Ammonia Borane | Dr. Susan Azpeitia, Dr. Claudio Mendicute-Fierro, Dr. Miguel A. Huertos, Dr. Antonio Rodríguez-Diéguez, Dr. José M. Seco, Dr. Antonio J. Mota, Prof. Dr. María A. Garralda. 2021. European Journal of Inorganic Chemistry. 2021: 879-891.
- Metal Ion-Driven Constitutional Adaptation in Dynamic Covalent C=C/C=N Organo-Metathesis | Dr. Ruirui Gu, Prof. Dr. Jean-Marie Lehn. 2021. Chemistry – An Asian Journal. 16: 44-48.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Quinoline-8-carbaldehyde, 1 g | sc-272135 | 1 g | $124.00 |
