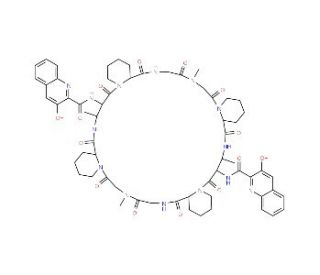
Quinaldopeptin (CAS 130743-07-6)
QUICK LINKS
Quinaldopeptin is a cyclic depsipeptide compound, initially isolated from the bacterium Streptomyces sp., known for its distinctive structural features, including a quinaldic acid moiety linked to a complex peptide ring. It functions primarily by interfering with bacterial RNA polymerase, a key enzyme in the transcription process, thereby inhibiting RNA synthesis. This action is achieved through binding to the beta subunit of RNA polymerase, an interaction that effectively blocks the transcription pathway necessary for bacterial growth and replication. In research, quinaldopeptin has been extensively used to study the mechanisms of transcription inhibition, particularly in prokaryotic organisms. Its unique mode of action makes it a valuable tool for probing the structure and function of RNA polymerase, as well as understanding the broader implications of transcriptional regulation in bacteria. Furthermore, the study of quinaldopeptin and its interactions has contributed to the field of microbial physiology by highlighting potential targets for the development of new inhibitors that can specifically disrupt bacterial transcription processes. This research is crucial for advancing knowledge of microbial resistance mechanisms and could help guide the design of novel compounds to combat resistant bacterial strains, thereby enriching the strategies available for controlling bacterial infections.
Quinaldopeptin (CAS 130743-07-6) References
- Structure of actinotetraose hexatiglate, a unique glucotetraose from an actinomycete bacterium. | Rickards, RW., et al. 1998. J Antibiot (Tokyo). 51: 1093-8. PMID: 10048568
- Bisintercalator natural products with potential therapeutic applications: isolation, structure determination, synthetic and biological studies. | Dawson, S., et al. 2007. Nat Prod Rep. 24: 109-26. PMID: 17268609
- Quinaldopeptin, a novel antibiotic of the quinomycin family. | Toda, S., et al. 1990. J Antibiot (Tokyo). 43: 796-808. PMID: 2387774
- Total synthesis of quinaldopeptin and its analogues. | Ichikawa, S., et al. 2013. J Org Chem. 78: 12662-70. PMID: 24236405
- Synthesis and biological evaluation of quinaldopeptin. | Katayama, K., et al. 2014. J Org Chem. 79: 2580-90. PMID: 24555872
- Kribbellichelins A and B, Two New Antibiotics from Kribbella sp. CA-293567 with Activity against Several Human Pathogens. | Virués-Segovia, JR., et al. 2022. Molecules. 27: PMID: 36234892
- Introduction of Novel Drug Targets against Staphylococcus aureus and Proposing Putative Inhibitors against Adenine N1 (m1A22)-tRNA Methyltransferase (TrmK) using Computer-aided Drug Discovery. | Beig, M., et al. 2023. Curr Pharm Des. 29: 1135-1147. PMID: 37132149
- Structural and functional analyses of the echinomycin resistance conferring protein Ecm16 from Streptomyces lasalocidi. | Gade, P., et al. 2023. Sci Rep. 13: 7980. PMID: 37198233
- Structure-Activity Relationships of Bis-Intercalating Peptides and Their Application as Antibody-Drug Conjugate Payloads. | Petersen, ME., et al. 2023. J Med Chem. 66: 8288-8309. PMID: 37307297
- Crystal Structure Characterization, Interaction Energy Analysis and DFT Studies of 3-(4-Chlorophenyl)-N-phenylquinoxalin-2-amine | Akhileshwari, P., et al. 2023. Journal of Chemical Crystallography. 53(2): 185-196.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Quinaldopeptin, 500 µg | sc-202304 | 500 µg | $440.00 |
