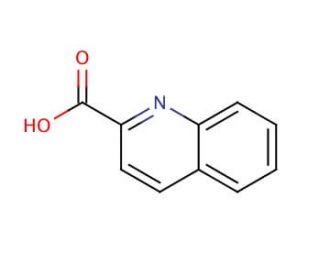
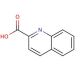
Quinaldic acid (CAS 93-10-7)
QUICK LINKS
Quinaldic acid functions as a chelating agent in various experimental applications. It acts by forming stable complexes with metal ions, particularly transition metals such as copper and iron. This chelation process involves the formation of coordinate covalent bonds between the quinaldic acid molecule and the metal ion, resulting in the formation of a stable complex. This property allows quinaldic acid to be utilized to sequester and stabilize metal ions, preventing their participation in unwanted chemical reactions. Quinaldic acid can also act as a ligand, binding to metal ions and influencing their reactivity in chemical reactions. Its ability to form stable complexes with metal ions may be a useful for various experimental procedures where the control and manipulation of metal ions is required.
Quinaldic acid (CAS 93-10-7) References
- The metabolism of quinaldic acid, kynurenic acid, and xanthurenic acid in the rabbit. | KAIHARA, M. and PRICE, JM. 1962. J Biol Chem. 237: 1727-9. PMID: 14453131
- Proton-transfer and non-transfer in compounds of quinoline and quinaldic acid with L-tartaric acid. | Smith, G., et al. 2006. Acta Crystallogr C. 62: o694-8. PMID: 17148915
- (Benzoato-κO,O')(quinoline-2-carboxyl-ato-κN,O)(quinoline-2-carboxylic acid-κN,O)manganese(II). | Martins, ND., et al. 2007. Acta Crystallogr Sect E Struct Rep Online. 64: m258. PMID: 21200595
- (Benzoato-κO,O')(quinoline-2-carboxyl-ato-κN,O)(quinoline-2-carboxylic acid-κN,O)copper(II). | Martins, ND., et al. 2008. Acta Crystallogr Sect E Struct Rep Online. 64: m829-30. PMID: 21202511
- Microwave-assisted synthesis of new substituted anilides of quinaldic acid. | Bobal, P., et al. 2012. Molecules. 17: 1292-306. PMID: 22293847
- Insights into quinaldic acid moiety formation in thiostrepton biosynthesis facilitating fluorinated thiopeptide generation. | Duan, L., et al. 2012. Chem Biol. 19: 443-8. PMID: 22520750
- Quinaldic acid inhibits proliferation of colon cancer ht-29 cells in vitro: effects on signaling pathways. | Langner, E., et al. 2015. Eur J Pharmacol. 757: 21-7. PMID: 25797283
- Thiostrepton Variants Containing a Contracted Quinaldic Acid Macrocycle Result from Mutagenesis of the Second Residue. | Zhang, F., et al. 2016. ACS Chem Biol. 11: 415-24. PMID: 26630475
- Quinaldic acid in synovial fluid of patients with rheumatoid arthritis and osteoarthritis and its effect on synoviocytes in vitro. | Nowicka-Stążka, P., et al. 2018. Pharmacol Rep. 70: 277-283. PMID: 29477035
- Quinaldic acid induces changes in the expression of p53 tumor suppressor both on protein and gene level in colon cancer LS180 cells. | Langner, E., et al. 2019. Pharmacol Rep. 71: 189-193. PMID: 30780127
- Insulin-releasing effect of quinaldic acid and its relatives on isolated Langerhans islets. | Okamoto, H., et al. 1973. Biochem Biophys Res Commun. 53: 1297-303. PMID: 4584024
- Mode of action of hypoglycemic agents. 3. Studies on 5-methoxy indole-2-carboxylic acid and quinaldic acid. | Reed, J. and Lardy, HA. 1970. J Biol Chem. 245: 5297-303. PMID: 5469167
- Effect of quinaldic acid and its relatives on insulin-release from isolated Langerhans islets. | Okamoto, H. 1975. Acta Vitaminol Enzymol. 29: 227-31. PMID: 801703
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Quinaldic acid, 5 g | sc-250824 | 5 g | $55.00 |
