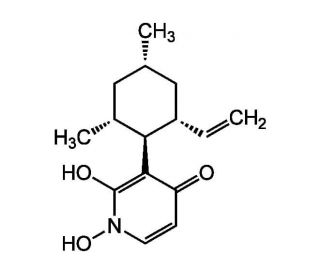
Molecular structure of Pyridoxatin, CAS Number: 135529-30-5
Pyridoxatin (CAS 135529-30-5)
Alternate Names:
Tolypocin
Application:
Pyridoxatin is a MMP-2 (Gelatinase A) inhibitor with antibiotic and anticancer properties
CAS Number:
135529-30-5
Purity:
≥95%
Molecular Weight:
263.33
Molecular Formula:
C15H21NO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Pyridoxatin, a fungal metabolite found in Acremonium, displays a wide range of biological activities, making it a versatile compound of research interest. With its free radical scavenging properties, it counteracts the damaging effects of free radicals, exhibiting an IC50 of 8 μM. As an inhibitor of matrix metalloproteinase-2 (MMP-2), it demonstrates its ability to impede the activity of this enzyme. Moreover, pyridoxatin showcases inhibitory effects on lipid peroxidation and DNA synthesis, further solidifying its multifaceted nature.
Pyridoxatin (CAS 135529-30-5) References
- Differential analysis of 2D NMR spectra: new natural products from a pilot-scale fungal extract library. | Schroeder, FC., et al. 2007. Angew Chem Int Ed Engl. 46: 901-4. PMID: 17183517
- Isolation and structural elucidation of pyridoxatin, a free radical scavenger of microbial origin. | Teshima, Y., et al. 1991. J Antibiot (Tokyo). 44: 685-7. PMID: 2071495
- Two new antibiotic pyridones produced by a marine fungus, Trichoderma sp. strain MF106. | Wu, B., et al. 2014. Mar Drugs. 12: 1208-19. PMID: 24663111
- Lichen endophyte derived pyridoxatin inactivates Candida growth by interfering with ergosterol biosynthesis. | Chang, W., et al. 2015. Biochim Biophys Acta. 1850: 1762-71. PMID: 25960388
- New PKS-NRPS tetramic acids and pyridinone from an Australian marine-derived fungus, Chaunopycnis sp. | Shang, Z., et al. 2015. Org Biomol Chem. 13: 7795-802. PMID: 26107107
- Tetramic Acids and Pyridone Alkaloids from the Endolichenic Fungus Tolypocladium cylindrosporum. | Li, XB., et al. 2015. J Nat Prod. 78: 2155-60. PMID: 26356746
- Establishing the Secondary Metabolite Profile of the Marine Fungus: Tolypocladium geodes sp. MF458 and Subsequent Optimisation of Bioactive Secondary Metabolite Production. | Kebede, B., et al. 2017. Mar Drugs. 15: PMID: 28333084
- Isolation and Characterization of Aphidicolin Derivatives from Tolypocladium inflatum. | Lin, J., et al. 2017. Molecules. 22: PMID: 28704971
- Antioxidant activity and cellular uptake of the hydroxamate-based fungal iron chelators pyridoxatin, desferriastechrome and desferricoprogen. | da Silva, GS., et al. 2019. Biometals. 32: 707-715. PMID: 31152280
- Screening of tenuazonic acid production-inducing compounds and identification of NPD938 as a regulator of fungal secondary metabolism. | Motoyama, T., et al. 2021. Biosci Biotechnol Biochem. 85: 2200-2208. PMID: 34379730
- Identification, Characterization, and Evaluation of Nematophagous Fungal Species of Arthrobotrys and Tolypocladium for the Management of Meloidogyne incognita. | Kassam, R., et al. 2021. Front Microbiol. 12: 790223. PMID: 34956156
- Lipopolysaccharide acting via toll-like receptor 4 transactivates the TGF-β receptor in vascular smooth muscle cells. | Afroz, R., et al. 2022. Cell Mol Life Sci. 79: 121. PMID: 35122536
- Liver-cell protective pyridones from the fungi Tolypocladium album dws120. | Wu, XQ., et al. 2023. Phytochemistry. 212: 113730. PMID: 37220864
- What was old is new again: Phenotypic screening of a unique fungal library yields pyridoxatin, a promising lead against extensively resistant Acinetobacter baumannii (AB5075). | Winter, HL., et al. 2023. Phytochem Lett. 55: 88-96. PMID: 37252254
Inhibitor of:
MMP-2.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pyridoxatin, 1 mg | sc-391043 | 1 mg | $163.00 |
