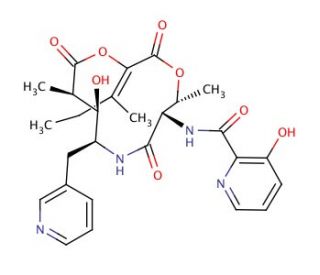
Pyridomycin (CAS 18791-21-4)
QUICK LINKS
Pyridomycin, a natural product isolated from Streptomyces pyridomyceticus, has attracted significant scientific interest due to its unique mechanisms of action and its role as a research tool in studying bacterial biochemistry and genetics. The compound specifically targets the bacterial enzyme DNA-dependent RNA polymerase, which is essential for RNA synthesis. Pyridomycin binds to this enzyme, thereby inhibiting the initiation phase of RNA transcription. This action is particularly valuable in studies focusing on transcriptional regulation and the identification of novel antibacterial targets. In scientific research, pyridomycin serves as a model compound to understand the interaction between small molecules and RNA polymerase. Structural studies, including X-ray crystallography and molecular docking, have been used extensively to explain the binding mode of pyridomycin, providing insights into the detailed molecular interactions that govern its inhibitory activity. These insights are crucial for the design of new inhibitors that mimic or enhance the action of pyridomycin.
Pyridomycin (CAS 18791-21-4) References
- Degradation studies on pyridomycin; chemical studies on antibiotics of Streptomyces. V. | MAEDA, K. 1957. J Antibiot (Tokyo). 10: 94-106. PMID: 13449017
- Identification and characterization of the pyridomycin biosynthetic gene cluster of Streptomyces pyridomyceticus NRRL B-2517. | Huang, T., et al. 2011. J Biol Chem. 286: 20648-57. PMID: 21454714
- Pyridomycin bridges the NADH- and substrate-binding pockets of the enoyl reductase InhA. | Hartkoorn, RC., et al. 2014. Nat Chem Biol. 10: 96-8. PMID: 24292073
- Functional Characterization of PyrG, an Unusual Nonribosomal Peptide Synthetase Module from the Pyridomycin Biosynthetic Pathway. | Huang, T., et al. 2016. Chembiochem. 17: 1421-5. PMID: 27197800
- Synthesis and Structure-Activity Relationship Studies of C2-Modified Analogs of the Antimycobacterial Natural Product Pyridomycin. | Kienle, M., et al. 2020. J Med Chem. 63: 1105-1131. PMID: 31904960
- Natural products for infectious microbes and diseases: an overview of sources, compounds, and chemical diversities. | Luo, L., et al. 2022. Sci China Life Sci. 65: 1123-1145. PMID: 34705221
- Characterization of Pyridomycin B Reveals the Formation of Functional Groups in Antimycobacterial Pyridomycin. | Huang, T., et al. 2022. Appl Environ Microbiol. 88: e0203521. PMID: 35108072
- Bifunctional NadC Homologue PyrZ Catalyzes Nicotinic Acid Formation in Pyridomycin Biosynthesis. | Zhou, Z., et al. 2023. ACS Chem Biol. 18: 141-150. PMID: 36517246
- A Novel Natural Siderophore Antibiotic Conjugate Reveals a Chemical Approach to Macromolecule Coupling. | Caradec, T., et al. 2023. ACS Cent Sci. 9: 2138-2149. PMID: 38033789
- The chemistry of pyridomycin. | Ogawara, H., et al. 1968. Chem Pharm Bull (Tokyo). 16: 679-87. PMID: 5677624
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pyridomycin, 500 µg | sc-397315 | 500 µg | $315.00 | |||
Pyridomycin, 2.5 mg | sc-397315A | 2.5 mg | $1100.00 |
