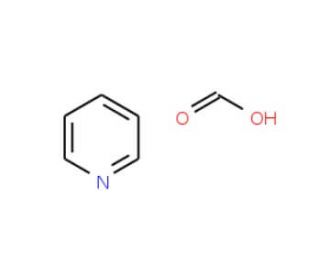
Molecular structure of Pyridinium formate buffer, CAS Number: 15066-28-1
Pyridinium formate buffer (CAS 15066-28-1)
Application:
Pyridinium formate buffer is a reagent for HPLC
CAS Number:
15066-28-1
Molecular Weight:
125.13
Molecular Formula:
C5H5N•CH2O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Pyridinium formate buffer is a chromatography reagent for HPLC. It has also been used to alter the electrical conductivity of solutions used during the synthesis of BiVO4 nanofibers for use as a photocatalyst for the removal of organic contaminants in impaired water sources. Pyridinium formate buffer is a versatile organic compound extensively utilized in scientific research for various purposes. This colorless and volatile compound readily dissolves in water, making it highly adaptable in laboratory applications. PIF serves as a reagent for synthesizing diverse organic compounds, a catalyst for facilitating chemical reactions, and a solvent for numerous biochemical processes.
Pyridinium formate buffer (CAS 15066-28-1) References
- Alkaloidal components in the poisonous plant, Ipomoea carnea (Convolvulaceae). | Haraguchi, M., et al. 2003. J Agric Food Chem. 51: 4995-5000. PMID: 12903959
- The enzymic synthesis of uridine diphosphate L-rhamnose. | BARBER, GA. 1962. Biochem Biophys Res Commun. 8: 204-8. PMID: 13864834
- On the sequence of residues 11 to 18 in bovine pancreatic ribonuclease. | SMYTH, DG., et al. 1962. J Biol Chem. 237: 1845-50. PMID: 13914531
- THE REDUCTION OF INORGANIC SULPHATE TO INORGANIC SULPHITE IN THE SMALL INTESTINE OF THE RAT. | ROBINSON, HC. 1965. Biochem J. 94: 687-91. PMID: 14340059
- On-line process monitoring of water-soluble ions in pulp and paper machine waters by capillary electrophoresis. | Kokkonen, R., et al. 2004. J Chromatogr A. 1032: 243-52. PMID: 15065802
- Speciation of cationic selenium compounds in Brassica juncea leaves by strong cation-exchange chromatography with inductively coupled plasma mass spectrometry. | Yathavakilla, SV., et al. 2005. J Chromatogr A. 1100: 153-9. PMID: 16198358
- Free and Protein-Bound Maillard Reaction Products in Beer: Method Development and a Survey of Different Beer Types. | Hellwig, M., et al. 2016. J Agric Food Chem. 64: 7234-43. PMID: 27594145
- Lysine-Derived Protein-Bound Heyns Compounds in Bakery Products. | Treibmann, S., et al. 2017. J Agric Food Chem. 65: 10562-10570. PMID: 29111707
- Determination of organic acids in seven wheat varieties by capillary gas chromatography. | Burke, DG., et al. 1985. Anal Biochem. 149: 421-9. PMID: 4073499
- Inhibition of mucopolysaccharide synthesis by 5-bromodeoxyuridine in cultures of chick amnion cells. | Mayne, R., et al. 1971. Dev Biol. 25: 547-67. PMID: 4256664
- Pyrimidine nucleosides. 6. Syntheses and anticancer activities of N4-substituted 2,2'-anhydronucleosides. | Kanai, T. and Ichino, M. 1974. J Med Chem. 17: 1076-8. PMID: 4529302
- Reactions of N-ethylmaleimide with peptides and amino acids. | Smyth, DG., et al. 1964. Biochem J. 91: 589-95. PMID: 5840721
- Des-lysyl glutamyl and des-lysyl pyroglutamyl ribonucleases. II. Structural studies. | Eaker, DL., et al. 1965. Biochemistry. 4: 1479-86. PMID: 5863315
- A comparison of the 16S ribosomal RNAs from mesophilic and thermophilic bacilli: some modifications in the Sanger method for RNA sequencing. | Woese, C., et al. 1976. J Mol Evol. 7: 197-213. PMID: 819656
- Human cells unable to express decoron produced disorganized extracellular matrix lacking 'shape modules' (interfibrillar proteoglycan bridges). | Scott, JE., et al. 1998. Exp Cell Res. 243: 59-66. PMID: 9716449
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pyridinium formate buffer, 100 ml | sc-296151 | 100 ml | $73.00 | |||
Pyridinium formate buffer, 500 ml | sc-296151A | 500 ml | $312.00 |
