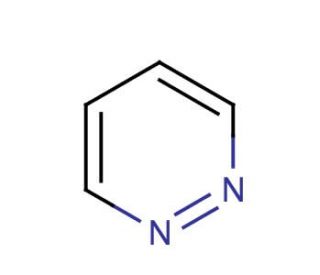
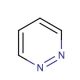
Pyridazine (CAS 289-80-5)
QUICK LINKS
Pyridazine is a heterocyclic aromatic compound. It consists of a pyridine nucleus fused with a 2-azabicyclo[2.2.2]octane ring. In the realm of scientific research, pyridazine plays a pivotal role in multiple domains. Pyridazine serves as a fundamental building block in organic synthesis, owing to its easy functionalization that allows for the formation of a variety of derivatives. Additionally, pyridazine acts as an intermediate in synthesizing diverse heterocyclic compounds such as pyridazines, pyridines, and pyrimidines. The precise mechanism of action of pyridazine remains not entirely elucidated. However, it is postulated that pyridazine may act as an inhibitor of enzymes involved in nucleic acid biosynthesis. Furthermore, pyridazine has demonstrated interactions with various proteins.
Pyridazine (CAS 289-80-5) References
- Bis[3,6-bis(6-methyl-2pyridyl)pyridazine-kappa2N2,N3]chlorocopper(II) perchlorate. | Choi, KY., et al. 2001. Acta Crystallogr C. 57: 47-8. PMID: 11173394
- Pharmacologically active pyridazine derivatives. Part 2. | Heinisch, G. and Kopelent-Frank, H. 1992. Prog Med Chem. 29: 141-83. PMID: 1475369
- Some recent approaches of biologically active substituted pyridazine and phthalazine drugs. | Asif, M. 2012. Curr Med Chem. 19: 2984-91. PMID: 22519394
- Strategy for the synthesis of pyridazine heterocycles and their derivatives. | Bel Abed, H., et al. 2013. J Org Chem. 78: 7845-58. PMID: 23947534
- Pyridazine Based Scaffolds as Privileged Structures in anti-Cancer Therapy. | Jaballah, MY., et al. 2017. Drug Res (Stuttg). 67: 138-148. PMID: 28073115
- Synthesis of Some New Pyridazine Derivatives for Anti-HAV Evaluation. | Flefel, EM., et al. 2017. Molecules. 22: PMID: 28106751
- Design and Synthesis of Pyridazine Containing Compounds with Promising Anticancer Activity. | Elmeligie, S., et al. 2017. Chem Pharm Bull (Tokyo). 65: 236-247. PMID: 28250345
- Pyridazine and pyridazinone derivatives as potent and selective factor XIa inhibitors. | Hu, Z., et al. 2018. Bioorg Med Chem Lett. 28: 987-992. PMID: 29501396
- Discovery of novel pyridazine derivatives as glucose transporter type 4 (GLUT4) translocation activators. | Tsuji, T., et al. 2019. Bioorg Med Chem Lett. 29: 1785-1790. PMID: 31101471
- New pyridazine derivatives as selective COX-2 inhibitors and potential anti-inflammatory agents; design, synthesis and biological evaluation. | Ahmed, EM., et al. 2020. Bioorg Chem. 95: 103497. PMID: 31838289
- Discovery of Novel Pyridazine-Based Cyclooxygenase-2 Inhibitors with a Promising Gastric Safety Profile. | Khan, A., et al. 2020. Molecules. 25: PMID: 32344801
- A One-Pot Approach to Novel Pyridazine C-Nucleosides. | Cermola, F., et al. 2021. Molecules. 26: PMID: 33920588
- [3 + n] Cycloaddition Reactions: A Milestone Approach for Elaborating Pyridazine of Potential Interest in Medicinal Chemistry and Optoelectronics. | Amariucai-Mantu, D., et al. 2021. Molecules. 26: PMID: 34199610
- Discovery of pyridachlometyl: A new class of pyridazine fungicides. | Manabe, A., et al. 2023. Bioorg Med Chem. 88-89: 117332. PMID: 37210791
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pyridazine, 5 g | sc-250812 | 5 g | $40.00 |
