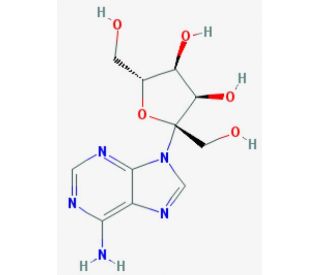
Psicofuranine (CAS 1874-54-0)
QUICK LINKS
Psicofuranine, with the CAS number 1874-54-0, is a nucleoside analog derived from the fermentation of Streptomyces hygroscopicus. It is recognized for its distinctive molecular structure featuring a furanose sugar linked to an adenine base. Psicofuranine acts primarily by inhibiting GMP synthetase, an essential enzyme in the biosynthesis of guanine nucleotides. This inhibition disrupts the production of GMP (guanosine monophosphate), a pivotal precursor in the synthesis of both GDP (guanosine diphosphate) and GTP (guanosine triphosphate), which are crucial for RNA synthesis and various cellular energy and signaling processes. In research contexts, psicofuranine has been extensively studied for its role in affecting purine metabolism, providing insights into cellular pathways involving nucleotide synthesis and utilization. These studies have helped delineate the mechanisms through which nucleoside analogs can influence genetic and metabolic activities in cells, contributing to a broader understanding of cellular regulation and metabolic control. Research on psicofuranine has also facilitated exploration into the dynamics of enzyme inhibition by nucleotide analogs, aiding in the development of models to predict the behavior of similar compounds in regulatory pathways. This makes psicofuranine a valuable tool in biochemical and genetic research, particularly in studies aimed at understanding and manipulating metabolic pathways for academic and applied sciences.
Psicofuranine (CAS 1874-54-0) References
- Plasmodium falciparum: isolation and characterisation of a gene encoding protozoan GMP synthase. | McConkey, GA. 2000. Exp Parasitol. 94: 23-32. PMID: 10631077
- Psicofuranine. VI. Antitumor and toxicopathological studies. | EVANS, JS. and GRAY, JE. 1959. Antibiot Chemother (Northfield). 9: 675-84. PMID: 13821001
- INHIBITION OF PARENTAL AND MUTANT XANTHOSINE 5'-PHOSPHATE AMINASES BY PSICOFURANINE. | UDAKA, S. and MOYED, HS. 1963. J Biol Chem. 238: 2797-803. PMID: 14063305
- [ACTION OF PSICOFURANINE ON HUMAN TUMORAL ELEMENTS IN CONTINUOUS CULTURE]. | DESSI, P., et al. 1963. Arch Ital Sci Farmacol. 13: 175-6. PMID: 14245136
- Role of purine biosynthetic intermediates in response to folate stress in Escherichia coli. | Rohlman, CE. and Matthews, RG. 1990. J Bacteriol. 172: 7200-10. PMID: 2254281
- Xanthosine-5'-phosphate amidotransferase from Escherichia coli. | Patel, N., et al. 1975. J Biol Chem. 250: 2609-13. PMID: 235520
- Nucleoside antibiotics: structure, biological activity, and biosynthesis. | Isono, K. 1988. J Antibiot (Tokyo). 41: 1711-39. PMID: 3061990
- Positional isotope exchange and kinetic experiments with Escherichia coli guanosine-5'-monophosphate synthetase. | von der Saal, W., et al. 1985. Biochemistry. 24: 5343-50. PMID: 3907701
- Formation of an adenyl xanthosine monophosphate intermediate by xanthosine 5'-phosphate aminase and its inhibition by psicofuranine. | Fukuyama, TT. 1966. J Biol Chem. 241: 4745-9. PMID: 5332732
- C4'-Branched-chain surgar nucleosides: synthesis of isomers of psicofuranine. | Rosenthal, A. and Ratcliffe, M. 1977. Carbohydr Res. 54: 61-73. PMID: 861968
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Psicofuranine, 1 mg | sc-202778 | 1 mg | $143.00 | |||
Psicofuranine, 5 mg | sc-202778A | 5 mg | $474.00 |
