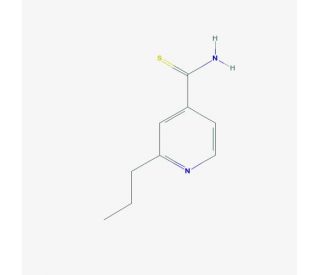
Prothionamide (CAS 14222-60-7)
QUICK LINKS
Prothionamide is a chemical compound that functions as an antibacterial agent in research and development applications. Its mechanism of action involves inhibiting the synthesis of mycolic acids in the cell wall of certain bacteria, specifically Mycobacterium tuberculosis. This disruption of mycolic acid synthesis leads to the impairment of cell wall formation and ultimately the death of the bacterial cells. Prothionamide achieves this by targeting the enzyme InhA, which is involved in the biosynthesis of mycolic acids. By interfering with this specific molecular target, prothionamide disrupts the structural integrity of the bacterial cell wall, rendering the bacteria more susceptible to other antibacterial agents.
Prothionamide (CAS 14222-60-7) References
- Hepatitis associated with prothionamide for treatment of multidrug-resistant tuberculosis. | Hsu, HL., et al. 2010. J Formos Med Assoc. 109: 923-7. PMID: 21195892
- Polymorphisms in isoniazid and prothionamide resistance genes of the Mycobacterium tuberculosis complex. | Projahn, M., et al. 2011. Antimicrob Agents Chemother. 55: 4408-11. PMID: 21709103
- Ex vivo conversion of prodrug prothionamide to its metabolite prothionamide sulfoxide with different extraction techniques and their estimation in human plasma by LC-MS/MS. | Trivedi, V., et al. 2013. Bioanalysis. 5: 185-200. PMID: 23330561
- Low Serum Concentrations of Moxifloxacin, Prothionamide, and Cycloserine on Sputum Conversion in Multi-Drug Resistant TB. | Lee, SH., et al. 2015. Yonsei Med J. 56: 961-7. PMID: 26069117
- Efficacy and tolerability of ethionamide versus prothionamide: a systematic review. | Scardigli, A., et al. 2016. Eur Respir J. 48: 946-52. PMID: 27288034
- Prothionamide susceptibility testing of Mycobacterium tuberculosis using the resazurin microtitre assay and the BACTECMGIT 960 system. | Tan, Y., et al. 2017. Eur J Clin Microbiol Infect Dis. 36: 779-782. PMID: 28000029
- Molecular Characterization of Prothionamide-Resistant Mycobacterium tuberculosis Isolates in Southern China. | Tan, Y., et al. 2017. Front Microbiol. 8: 2358. PMID: 29250048
- Development and evaluation of Chitosan nanoparticles based dry powder inhalation formulations of Prothionamide. | Debnath, SK., et al. 2018. PLoS One. 13: e0190976. PMID: 29370192
- Mutation EthAW21R confers co-resistance to prothionamide and ethionamide in both Mycobacterium bovis BCG and Mycobacterium tuberculosis H37Rv. | Mugweru, J., et al. 2018. Infect Drug Resist. 11: 891-894. PMID: 29942141
- Bacterial Genome-Wide Association Identifies Novel Factors That Contribute to Ethionamide and Prothionamide Susceptibility in Mycobacterium tuberculosis. | Hicks, ND., et al. 2019. mBio. 10: PMID: 31015328
- Development and validation of a simple LC-MS/MS method for simultaneous determination of moxifloxacin, levofloxacin, prothionamide, pyrazinamide and ethambutol in human plasma. | Zheng, X., et al. 2020. J Chromatogr B Analyt Technol Biomed Life Sci. 1158: 122397. PMID: 33091676
- Identification and characterization of Prothionamide degradation impurities by mass spectrometry, NMR spectroscopy, and ultra high performance liquid chromatography method development. | Baksam, VK., et al. 2021. J Sep Sci. 44: 2078-2088. PMID: 33733566
- Ethionamide and Prothionamide Based Coumarinyl-Thiazole Derivatives: Synthesis, Antitubercular Activity, Toxicity Investigations and Molecular Docking Studies. | Imran, M. 2022. Pharm Chem J. 56: 1215-1225. PMID: 36531826
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Prothionamide, 1 g | sc-204865 | 1 g | $47.00 | |||
Prothionamide, 5 g | sc-204865A | 5 g | $92.00 |
